Participating in Health Research Studies
What is health research.
- Is Health Research Safe?
- Is Health Research Right for Me?
- Types of Health Research
The term "health research," sometimes also called "medical research" or "clinical research," refers to research that is done to learn more about human health. Health research also aims to find better ways to prevent and treat disease. Health research is an important way to help improve the care and treatment of people worldwide.
Have you ever wondered how certain drugs can cure or help treat illness? For instance, you might have wondered how aspirin helps reduce pain. Well, health research begins with questions that have not been answered yet such as:
"Does a certain drug improve health?"
To gain more knowledge about illness and how the human body and mind work, volunteers can help researchers answer questions about health in studies of an illness. Studies might involve testing new drugs, vaccines, surgical procedures, or medical devices in clinical trials . For this reason, health research can involve known and unknown risks. To answer questions correctly, safely, and according to the best methods, researchers have detailed plans for the research and procedures that are part of any study. These procedures are called "protocols."
An example of a research protocol includes the process for determining participation in a study. A person might meet certain conditions, called "inclusion criteria," if they have the required characteristics for a study. A study on menopause may require participants to be female. On the other hand, a person might not be able to enroll in a study if they do not meet these criteria based on "exclusion criteria." A male may not be able to enroll in a study on menopause. These criteria are part of all research protocols. Study requirements are listed in the description of the study.

A Brief History
While a few studies of disease were done using a scientific approach as far back as the 14th Century, the era of modern health research started after World War II with early studies of antibiotics. Since then, health research and clinical trials have been essential for the development of more than 1,000 Food and Drug Administration (FDA) approved drugs. These drugs help treat infections, manage long term or chronic illness, and prolong the life of patients with cancer and HIV.
Sound research demands a clear consent process. Public knowledge of the potential abuses of medical research arose after the severe misconduct of research in Germany during World War II. This resulted in rules to ensure that volunteers freely agree, or give "consent," to any study they are involved in. To give consent, one should have clear knowledge about the study process explained by study staff. Additional safeguards for volunteers were also written in the Nuremberg Code and the Declaration of Helsinki .
New rules and regulations to protect research volunteers and to eliminate ethical violations have also been put in to place after the Tuskegee trial . In this unfortunate study, African American patients with syphilis were denied known treatment so that researchers could study the history of the illness. With these added protections, health research has brought new drugs and treatments to patients worldwide. Thus, health research has found cures to many diseases and helped manage many others.
Why is Health Research Important?
The development of new medical treatments and cures would not happen without health research and the active role of research volunteers. Behind every discovery of a new medicine and treatment are thousands of people who were involved in health research. Thanks to the advances in medical care and public health, we now live on average 10 years longer than in the 1960's and 20 years longer than in the 1930's. Without research, many diseases that can now be treated would cripple people or result in early death. New drugs, new ways to treat old and new illnesses, and new ways to prevent diseases in people at risk of developing them, can only result from health research.
Before health research was a part of health care, doctors would choose medical treatments based on their best guesses, and they were often wrong. Now, health research takes the guesswork out. In fact, the Food and Drug Administration (FDA) requires that all new medicines are fully tested before doctors can prescribe them. Many things that we now take for granted are the result of medical studies that have been done in the past. For instance, blood pressure pills, vaccines to prevent infectious diseases, transplant surgery, and chemotherapy are all the result of research.
Medical research often seems much like standard medical care, but it has a distinct goal. Medical care is the way that your doctors treat your illness or injury. Its only purpose is to make you feel better and you receive direct benefits. On the other hand, medical research studies are done to learn about and to improve current treatments. We all benefit from the new knowledge that is gained in the form of new drugs, vaccines, medical devices (such as pacemakers) and surgeries. However, it is crucial to know that volunteers do not always receive any direct benefits from being in a study. It is not known if the treatment or drug being studied is better, the same, or even worse than what is now used. If this was known, there would be no need for any medical studies.
- Next: Is Health Research Safe? >>
- Last Updated: May 27, 2020 3:05 PM
- URL: https://guides.library.harvard.edu/healthresearch
Medical Dictionary
Search medical terms and abbreviations with the most up-to-date and comprehensive medical dictionary from the reference experts at Merriam-Webster. Master today's medical vocabulary. Become an informed health-care consumer!
Browse the Medical Dictionary
Featured game.

Find the Best Synonym
Test your vocabulary with our 10-question quiz!
Deniz Burnham & 'Astronaut'
Peyton Manning & 'Omaha'
Issa Rae & 'Insecure'
Word of the day.
See Definitions and Examples »
Get Word of the Day daily email!
Games & Quizzes

Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
Medical research articles from across Nature Portfolio
Medical research involves research in a wide range of fields, such as biology, chemistry, pharmacology and toxicology with the goal of developing new medicines or medical procedures or improving the application of those already available. It can be viewed as encompassing preclinical research (for example, in cellular systems and animal models) and clinical research (for example, clinical trials).
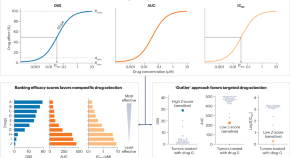
Functional precision medicine for pediatric cancers
A small, prospective clinical study shows that ex vivo drug screening of pediatric cancer samples can identify effective therapeutic options. If validated, these findings could herald a new approach to precision medicine in this setting.
- M. Emmy M. Dolman
- Paul G. Ekert
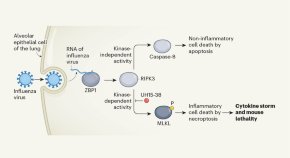
Blocking cell death limits lung damage and inflammation from influenza
Animals that receive an inhibitor of an antiviral cell-death response called necroptosis are less likely to die of influenza even at a late stage of infection. This has implications for the development of therapies for respiratory diseases.
- Nishma Gupta
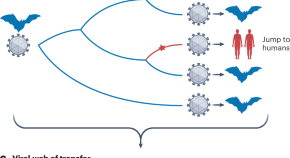
Frequent jumps from human hosts
An analysis of publicly available viral genomes explores the evolutionary dynamics of host jumps and shows that humans are as much a source of viral spillover events to other animals as they are recipients.
- Christophe Dessimoz
Related Subjects
- Drug development
- Epidemiology
- Experimental models of disease
- Genetics research
- Outcomes research
- Paediatric research
- Preclinical research
- Stem-cell research
- Clinical trial design
- Translational research
Latest Research and Reviews
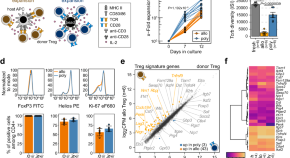
Donor regulatory T cells rapidly adapt to recipient tissues to control murine acute graft-versus-host disease
Graft-versus-Host disease is a major complication after allogeneic bone marrow transplantation and is ameliorated by adoptively transferred donor regulatory T cells. Here, the authors apply transcriptomic and TCR profiling to assess regulatory T cell organ-specific adaptation in murine bone marrow transplantation models.
- David J. Dittmar
- Franziska Pielmeier
- Michael Rehli
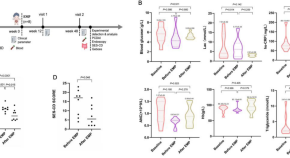
Empagliflozin in children with glycogen storage disease-associated inflammatory bowel disease: a prospective, single-arm, open-label clinical trial
- Xiaoyan Zhang
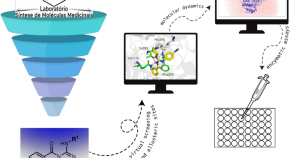
Molecular modelling studies and in vitro enzymatic assays identified A 4-(nitrobenzyl)guanidine derivative as inhibitor of SARS-CoV-2 Mpro
- Kaio Maciel de Santiago-Silva
- Priscila Goes Camargo
- Marcelle de Lima Ferreira Bispo
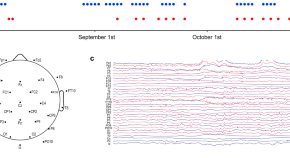
Open access EEG dataset of repeated measurements from a single subject for microstate analysis
- Shuyong Jia
- Guangjun Wang
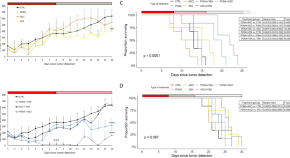
Combination therapies with ponatinib and asciminib in a preclinical model of chronic myeloid leukemia blast crisis with compound mutations
- Nikola Curik
- Adam Laznicka
- Katerina Machova Polakova
Prediction of hematologic toxicity in luminal type breast cancer patients receiving neoadjuvant chemotherapy using CT L1 level skeletal muscle index
- Pinxiu Wang
- Wenzhen Yuan
News and Comment
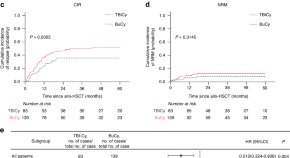
Busulfan plus cyclophosphamide vs. total body irradiation plus cyclophosphamide for allogeneic hematopoietic stem cell transplantation in patients with acute T lymphoblastic leukemia: a large-scale propensity score matching-based study
- Minyuan Liu
Safety and efficacy of a new academic CD19-directed CAR-T cell for refractory/relapsed non-Hodgkin lymphoma and acute lymphoblastic leukemia in Brazil
- Camila D. Donadel
- Gil C. De Santis
- Diego V. Clé
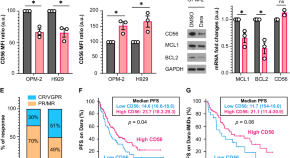
CD56 expression predicts response to Daratumumab-based regimens
- Allen J. Robinette
- Laila Huric
- Francesca Cottini
Efficacy of eculizumab in transplantation-associated thrombotic microangiopathy: results of the French nationwide study on behalf of the SFGM-TC and the CNR-MAT
- Marion Peyre
- Flore Sicre de Fontbrune

Why diversity is needed at every level of clinical trials, from participants to leaders
Diversity in clinical trials must be accompanied by justice and equity, including benefits for underrepresented participants, in order to boost population health.
- Khadijah Breathett
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
An official website of the United States government
The .gov means it's official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you're on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- Browse Titles
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Institute of Medicine (US) Clinical Research Roundtable; Tunis S, Korn A, Ommaya A, editors. The Role of Purchasers and Payers in the Clinical Research Enterprise: Workshop Summary. Washington (DC): National Academies Press (US); 2002.

The Role of Purchasers and Payers in the Clinical Research Enterprise: Workshop Summary.
- Hardcopy Version at National Academies Press
Appendix V Definitions of Clinical Research and Components of the Enterprise
- DEFINITION OF CLINICAL RESEARCH
(Clinical Research: A National Call to Action, November 1999) Clinical research is a component of medical and health research intended to produce knowledge valuable for understanding human disease, preventing and treating illness, and promoting health. Clinical Research embraces a continuum of studies involving interactions with patients, diagnostic clinical materials or data, or populations in any of the following categories: (1) disease mechanisms (etiopathogenesis); (2) bi-directional integrative (translational) research; (3) clinical knowledge, detection, diagnosis and natural history of disease; (4) therapeutic interventions including development and clinical trials of drugs, biologics, devices, and instruments; (5) prevention (primary and secondary) and health promotion; (6) behavioral research; (7) health services research, including outcomes, and cost-effectiveness; (8) epidemiology; and (9) community-based and managed care-based trials.
- MAJOR COMPONENTS OF THE CLINICAL RESEARCH ENTERPRISE
Sponsors include private and public sector funding organizations such as the National Institutes of Health, pharmaceutical companies, medical device manufacturers, biotechnology firms, universities, private foundations, and national societies. Within the public sector the National Institutes of Health (NIH) is the largest clinical research sponsor, followed by the Department of Defense (DOD), the Department of Veterans Affairs (VA), Agency for Healthcare Research and Quality (AHRQ), and the Centers for Disease Control (CDC).
Research Organizations
Research organizations include academic health centers, private research institutes, survey research organizations, federal government intramural research programs, and contract research organizations.
Investigators
Investigators are the scientists performing clinical research from varied disciplines with a range of academic qualifications (e.g., MD, Ph.D., RN, DDS, PharmD).
Participants
Participants are the human volunteers, medical information and biological materials of human origin, or data derived from volunteers. Participants may have particular health conditions or may be healthy volunteers or populations at large.
Oversight Entities
Oversight entities include Institutional Review Boards, Food and Drug Administration, Department of Health and Human Services, Veterans Affairs, National Committee for Quality Assurance, and other national regulatory agencies.
Stakeholders/Consumers
Stakeholders/Consumers include health insurers, managed care organizations, health care systems, organized medicine, voluntary health agencies, patient advocacy groups, purchasers of health care, and providers of health care, public health systems, and individual consumers.
- Cite this Page Institute of Medicine (US) Clinical Research Roundtable; Tunis S, Korn A, Ommaya A, editors. The Role of Purchasers and Payers in the Clinical Research Enterprise: Workshop Summary. Washington (DC): National Academies Press (US); 2002. Appendix V, Definitions of Clinical Research and Components of the Enterprise.
- PDF version of this title (1.0M)
In this Page
Recent activity.
- Definitions of Clinical Research and Components of the Enterprise - The Role of ... Definitions of Clinical Research and Components of the Enterprise - The Role of Purchasers and Payers in the Clinical Research Enterprise
Your browsing activity is empty.
Activity recording is turned off.
Turn recording back on
Connect with NLM
National Library of Medicine 8600 Rockville Pike Bethesda, MD 20894
Web Policies FOIA HHS Vulnerability Disclosure
Help Accessibility Careers
- U.S. Department of Health & Human Services

- Virtual Tour
- Staff Directory
- En Español

Autism Spectrum Disorder
Learn about NIH-supported research on autism spectrum disorder, including improved early screening and diagnosis.
Learn more »

Taking Care of Your Voice
Does your job put great demands on your voice? You may be at risk for developing voice problems. Find ways to protect your voice.

Supporting Addiction Research for 50 Years
The National Institute on Drug Abuse (NIDA) supports scientific research to improve the understanding, prevention, & treatment of drug use & addiction.

Alcohol Awareness
Take time to reflect on the role alcohol plays in your life.

Take the Virtual Tour
Explore the Bethesda campus and how NIH turns discovery into health.
In the News


Rare Disease Research
Clinical trial could lead to first effective treatment for ACDC disease.

Polycystic Kidney Disease
New disease modeling and gene editing techniques shed light on this common ailment.

Neurodegenerative Diseases
A simple skin biopsy could identify people with diseases like Parkinson’s.

Gene Therapy
Experimental treatment for rare childhood condition is safe, slows progression.
NIH at a Glance
Virtual-tour-screenshot-square.jpg.

monica-bertagnolli-thumbnail.jpg

The NIH Director
Monica M. Bertagnolli, M.D., is the NIH Director and provides leadership for the 27 Institutes and Centers that make up NIH.
nih-at-a-glance-funding.jpg
Funding for Research
NIH is the largest source of funding for medical research in the world, creating hundreds of thousands of high-quality jobs.
nih-at-a-glance-labs.jpg

Labs at NIH
Scientists conduct research on NIH campuses across the U.S., as part of our Intramural Research Program.
improving-health-collage.jpg

Impact of NIH Research
NIH-supported research has had a major positive impact on nearly all of our lives.
researcher-holding-petri-dish.jpg

Jobs at NIH
The central recruitment point of access to all NIH jobs and training opportunities
A public-private partnership to develop a coordinated research strategy to speed the most promising COVID-19 vaccines and treatments.
Rapid Acceleration of Diagnostics (RADx)
An initiative to speed innovation in the development, commercialization, and implementation of technologies for COVID-19 testing.
A new research initiative to understand, prevent, and treat the long-term effects of COVID-19.
COVID-19 Treatment Guidelines
Guidelines from NIH for the diagnosis, treatment, and control of COVID-19.
Research information from NIH
NIH supports research in COVID-19 testing, treatments, and vaccines. También disponible en español.
NIH COVID-19 Safety Plan
Guidance to NIH staff, including employees, contractors, trainees, and volunteers, related to COVID-19.
Featured Resources & Initiatives
A new science agency proposed by President Joseph Biden as part of NIH to drive biomedical breakthroughs and provide transformative solutions for all patients.
Anti-Sexual Harassment
NIH does not tolerate pervasive or severe harassment of any kind, including sexual harassment.
Ending Structural Racism
Learn more about NIH’s efforts to end structural racism in biomedical research through the UNITE initiative.
All of Us Research Program
A research effort to revolutionize how we improve health and treat disease.
NIH HEAL Initiative
Trans-agency effort to speed scientific solutions to stem the national opioid crisis.
Clinical Trials
Learn about participating in clinical trials and where to find them.
Accelerating Medicines Partnership
A bold venture to help identify new treatments and cures for diseases.
Medical Research Initiatives
Important initiatives aimed at improving medical research.
Training at NIH
NIH provides training opportunities internally, as well as at universities and other institutions across the U.S.
Connect with Us
- More Social Media from NIH
- ABBREVIATIONS
- BIOGRAPHIES
- CALCULATORS
- CONVERSIONS
- DEFINITIONS

Vocabulary
What does medical research mean?
Definitions for medical research med·ical re·search, this dictionary definitions page includes all the possible meanings, example usage and translations of the word medical research ., wikipedia rate this definition: 0.0 / 0 votes.
Medical research
Biomedical research (or experimental medicine) encompasses a wide array of research, extending from "basic research" (also called bench science or bench research), – involving fundamental scientific principles that may apply to a preclinical understanding – to clinical research, which involves studies of people who may be subjects in clinical trials. Within this spectrum is applied research, or translational research, conducted to expand knowledge in the field of medicine. Both clinical and preclinical research phases exist in the pharmaceutical industry's drug development pipelines, where the clinical phase is denoted by the term clinical trial. However, only part of the clinical or preclinical research is oriented towards a specific pharmaceutical purpose. The need for fundamental and mechanism-based understanding, diagnostics, medical devices, and non-pharmaceutical therapies means that pharmaceutical research is only a small part of medical research. The increased longevity of humans over the past century can be significantly attributed to advances resulting from medical research. Among the major benefits of medical research have been vaccines for measles and polio, insulin treatment for diabetes, classes of antibiotics for treating a host of maladies, medication for high blood pressure, improved treatments for AIDS, statins and other treatments for atherosclerosis, new surgical techniques such as microsurgery, and increasingly successful treatments for cancer. New, beneficial tests and treatments are expected as a result of the Human Genome Project. Many challenges remain, however, including the appearance of antibiotic resistance and the obesity epidemic. Most of the research in the field is pursued by biomedical scientists, but significant contributions are made by other type of biologists. Medical research on humans, has to strictly follow the medical ethics sanctioned in the Declaration of Helsinki and hospital review board where the research is conducted. In all cases, research ethics are expected.
Wikidata Rate this definition: 0.0 / 0 votes
Biomedical research, in general simply known as medical research, is the basic research, applied research, or translational research conducted to aid and support the body of knowledge in the field of medicine. Medical research can be divided into two general categories: the evaluation of new treatments for both safety and efficacy in what are termed clinical trials, and all other research that contributes to the development of new treatments. The latter is termed preclinical research if its goal is specifically to elaborate knowledge for the development of new therapeutic strategies. A new paradigm to biomedical research is being termed translational research, which focuses on iterative feedback loops between the basic and clinical research domains to accelerate knowledge translation from the bedside to the bench, and back again. Medical research may involve doing research on public health, biochemistry, clinical research, microbiology, physiology, oncology, surgery and research on many other non-communicable diseases such as diabetes and cardiovascular diseases. The increased longevity of humans over the past century can be significantly attributed to advances resulting from medical research. Among the major benefits have been vaccines for measles and polio, insulin treatment for diabetes, classes of antibiotics for treating a host of maladies, medication for high blood pressure, improved treatments for AIDS, statins and other treatments for atherosclerosis, new surgical techniques such as microsurgery, and increasingly successful treatments for cancer. New, beneficial tests and treatments are expected as a result of the Human Genome Project. Many challenges remain, however, including the appearance of antibiotic resistance and the obesity epidemic.
How to pronounce medical research?
Alex US English David US English Mark US English Daniel British Libby British Mia British Karen Australian Hayley Australian Natasha Australian Veena Indian Priya Indian Neerja Indian Zira US English Oliver British Wendy British Fred US English Tessa South African
How to say medical research in sign language?
Chaldean Numerology
The numerical value of medical research in Chaldean Numerology is: 2
Pythagorean Numerology
The numerical value of medical research in Pythagorean Numerology is: 7
Examples of medical research in a Sentence
Huseyin Naci :
According to our previous research, there is a bias against testing exercise, medical research increasingly favors drug interventions over strategies to modify lifestyle.
Senate Committee on Commerce :
Sharks and the scientists who study them have led us innovations in improvements in aerodynamics, renewable energy, electrical sensors, and health and medical research . Innovative shark research can benefit marine ecosystems and continue to raise public awareness about these important species.
Democratic Representative Rosa DeLauro :
Pfizer built their business on the back of our research and development tax incentives, our federally supported medical research , our skilled workforce, and our infrastructure, we cannot continue to allow Pfizer and other corporations to pretend that they are American while reaping the benefits this country has to offer, yet claiming to be another nationality when the tax bill comes.
Francis Collins :
I have done everything I can to stay out of any kind of political, partisan debates because it is really not a place where medical research belongs, i was not going to compromise scientific principles to just hold onto the job.
Bill Evanina :
Medical research organizations and those who work for them should be vigilant against threat actors seeking to steal intellectual property or other sensitive data related to America's response to the COVID19 pandemic.
- ^ Wikipedia https://en.wikipedia.org/wiki/Medical_Research
- ^ Wikidata https://www.wikidata.org/w/index.php?search=medical research
Translations for medical research
From our multilingual translation dictionary.
- بحث طبى Arabic
Word of the Day
Would you like us to send you a free new word definition delivered to your inbox daily.
Please enter your email address:
Citation
Use the citation below to add this definition to your bibliography:.
Style: MLA Chicago APA
"medical research." Definitions.net. STANDS4 LLC, 2024. Web. 15 Apr. 2024. < https://www.definitions.net/definition/medical+research >.
Discuss these medical research definitions with the community:
Report Comment
We're doing our best to make sure our content is useful, accurate and safe. If by any chance you spot an inappropriate comment while navigating through our website please use this form to let us know, and we'll take care of it shortly.
You need to be logged in to favorite .
Create a new account.
Your name: * Required
Your email address: * Required
Pick a user name: * Required
Username: * Required
Password: * Required
Forgot your password? Retrieve it
Are we missing a good definition for medical research ? Don't keep it to yourself...
Image credit, the web's largest resource for, definitions & translations, a member of the stands4 network, image or illustration of.
- medical research

Free, no signup required :
Add to chrome, add to firefox, browse definitions.net, are you a words master, the transparent covering of an aircraft cockpit, Nearby & related entries:.
- medical referral source
- medical regulating
- medical reimbursements of america
- medical relation noun
- medical report noun
- medical savings account
- medical savings accounts
- medical school noun
- medical science noun
- medical scientist noun
Alternative searches for medical research :
- Search for medical research on Amazon


IMAGES
VIDEO
COMMENTS
The University of Florida Cancer and Genetics Research Complex is an integrated medical research facility. Medical research (or biomedical research ), also known as health research, refers to the process of using scientific methods with the aim to produce knowledge about human diseases, the prevention and treatment of illness, and the promotion ...
re·search. ( rēsĕrch, rē-sĕrch) 1. The organized quest for new knowledge and better understanding (e.g., of the natural world or determinants of health and disease). Five types of research are recognized: observational (empiric), analytic, experimental, theoretic, applied. 2. To conduct such scientific inquiry.
Clinical Research. Clinical research is medical research that involves people to test new treatments and therapies. Clinical Trial. A research study in which one or more human subjects are prospectively assigned to one or more interventions (which may include placebo or other control) to evaluate the effects of those interventions on health-related biomedical or behavioral outcomes.
medical research. Research concerned with any phase of medical science. Want to thank TFD for its existence? Tell a friend about us, add a link to this page, or visit the webmaster's page for free fun content . Looking for online definition of medical research in the Medical Dictionary? medical research explanation free. What is medical research?
Clinical research is an alternative terminology used to describe medical research. Clinical research involves people, and it is generally carried out to evaluate the efficacy of a therapeutic drug, a medical/surgical procedure, or a device as a part of treatment and patient management. ... Definition: Randomized trial: Study participants are ...
Research Dictionary Are we missing a clinical research term or acronym? Let us ... Term or Acronym Definition Context; A-110: OMB Circular A-110 - Uniform Administrative Requirements for Grants and Agreements with Institutions of Higher Education, Hospitals, or Other Nonprofit Organizations ... United States Army Medical Research and Materiel ...
Clinical research is the comprehensive study of the safety and effectiveness of the most promising advances in patient care. Clinical research is different than laboratory research. It involves people who volunteer to help us better understand medicine and health. Lab research generally does not involve people — although it helps us learn ...
Medical research often seems much like standard medical care, but it has a distinct goal. Medical care is the way that your doctors treat your illness or injury. Its only purpose is to make you feel better and you receive direct benefits. On the other hand, medical research studies are done to learn about and to improve current treatments.
Define medical research. medical research synonyms, medical research pronunciation, medical research translation, English dictionary definition of medical research. n. 1. Careful study of a given subject, field, or problem, undertaken to discover facts or principles. 2. An act or period of such study: her researches of...
Cohort studies in a population group in which there has been exposure (e.g. industrial workers) Study of multiple exposures, such as the combined effect of oral contraceptives and smoking on myocardial infarction. Case control studies. Study of multiple end points, such as mortality from different causes.
A procedure designed to provide insight into the structure of a clinical problem and to identify the main determinants of diagnostic and therapeutic choice. This procedure is useful to small numbers of clinical cases, even to a single patient (see n -of-1 study). The procedure has four stages: 1.
Medical Dictionary. Search medical terms and abbreviations with the most up-to-date and comprehensive medical dictionary from the reference experts at Merriam-Webster. Master today's medical vocabulary. Become an informed health-care consumer!
Adverse Event (AE) - Any untoward or unfavorable medical occurrence in a clinical research study participant, including any abnormal sign (e.g. abnormal physical exam or laboratory finding), symptom, or disease, temporally associated with the participants' involvement in the research, whether or not considered related to participation in the research.
Abstract. 1 Basics of Medical Research Research in any field is an enterprise that carries its own risks and benefits. One may make heavy investment in terms of time, money and expertise yet the ...
The Medical Research Involving Human Subjects Act (WMO) gives no clear definition. The law defines 'scientific research' as: 'medical research, part of which involves subjecting persons to actions or imposing certain behaviours upon them'. The explanatory memorandum1 also speaks of 'research with the goal of developing or improving ...
Definition. Medical research involves research in a wide range of fields, such as biology, chemistry, pharmacology and toxicology with the goal of developing new medicines or medical procedures or ...
Medical Dictionary is intended for use by healthcare consumers, students, and professionals as well as anyone who wants to keep up with the burgeoning array of terminology found in today's medical news. By staying clear of jargon, the dictionary offers fast and concise information, whether the user is searching for a description of an over-the-counter or prescription medication, a medical ...
(Clinical Research: A National Call to Action, November 1999) Clinical research is a component of medical and health research intended to produce knowledge valuable for understanding human disease, preventing and treating illness, and promoting health. Clinical Research embraces a continuum of studies involving interactions with patients, diagnostic clinical materials or data, or populations ...
Official website of the National Institutes of Health (NIH). NIH is one of the world's foremost medical research centers. An agency of the U.S. Department of Health and Human Services, the NIH is the Federal focal point for health and medical research. The NIH website offers health information for the public, scientists, researchers, medical professionals, patients, educators, and students ...
Definition of medical research in the Definitions.net dictionary. Meaning of medical research. What does medical research mean? ... Find a translation for the medical research definition in other languages: Select another language: - Select - 简体中文 (Chinese - Simplified) 繁體中文 (Chinese - Traditional)
The overall goal of a medical researcher is to improve scientific knowledge on topics related to medicine and public health. This includes completing biomedical research on topics like bacteria, viruses, pathogens and other infectious things. They may work with physicians, health departments or similar professionals to develop public health ...
1. The organized quest for new knowledge and better understanding (e.g., of the natural world or determinants of health and disease). Five types of research are recognized: observational (empiric), analytic, experimental, theoretic, applied. 2. To conduct such scientific inquiry.
Define Medical research. means studies and experiments done to increase or verify information about physical or mental diseases and disabilities and their causes, diagnosis, prevention, treatment, or control. The organization must conduct the research directly and continuously. If it primarily gives funds to other organizations (or grants and scholarships to individuals) for them to do the ...
Voids, or empty spaces, exist within matter at all scales, from the astronomical to the microscopic. In a new study, researchers used high-powered microscopy and mathematical theory to unveil ...