How-to conduct a systematic literature review: A quick guide for computer science research
Affiliations.
- 1 Faculty of Engineering, Mondragon University.
- 2 Design Innovation Center(DBZ), Mondragon University.
- PMID: 36405369
- PMCID: PMC9672331
- DOI: 10.1016/j.mex.2022.101895
Performing a literature review is a critical first step in research to understanding the state-of-the-art and identifying gaps and challenges in the field. A systematic literature review is a method which sets out a series of steps to methodically organize the review. In this paper, we present a guide designed for researchers and in particular early-stage researchers in the computer-science field. The contribution of the article is the following:•Clearly defined strategies to follow for a systematic literature review in computer science research, and•Algorithmic method to tackle a systematic literature review.
Keywords: Systematic literature reviews; computer science; doctoral studies; literature reviews; research methodology.
© 2022 The Author(s).
- Open access
- Published: 14 August 2018

Defining the process to literature searching in systematic reviews: a literature review of guidance and supporting studies
- Chris Cooper ORCID: orcid.org/0000-0003-0864-5607 1 ,
- Andrew Booth 2 ,
- Jo Varley-Campbell 1 ,
- Nicky Britten 3 &
- Ruth Garside 4
BMC Medical Research Methodology volume 18 , Article number: 85 ( 2018 ) Cite this article
205k Accesses
206 Citations
117 Altmetric
Metrics details
Systematic literature searching is recognised as a critical component of the systematic review process. It involves a systematic search for studies and aims for a transparent report of study identification, leaving readers clear about what was done to identify studies, and how the findings of the review are situated in the relevant evidence.
Information specialists and review teams appear to work from a shared and tacit model of the literature search process. How this tacit model has developed and evolved is unclear, and it has not been explicitly examined before.
The purpose of this review is to determine if a shared model of the literature searching process can be detected across systematic review guidance documents and, if so, how this process is reported in the guidance and supported by published studies.
A literature review.
Two types of literature were reviewed: guidance and published studies. Nine guidance documents were identified, including: The Cochrane and Campbell Handbooks. Published studies were identified through ‘pearl growing’, citation chasing, a search of PubMed using the systematic review methods filter, and the authors’ topic knowledge.
The relevant sections within each guidance document were then read and re-read, with the aim of determining key methodological stages. Methodological stages were identified and defined. This data was reviewed to identify agreements and areas of unique guidance between guidance documents. Consensus across multiple guidance documents was used to inform selection of ‘key stages’ in the process of literature searching.
Eight key stages were determined relating specifically to literature searching in systematic reviews. They were: who should literature search, aims and purpose of literature searching, preparation, the search strategy, searching databases, supplementary searching, managing references and reporting the search process.
Conclusions
Eight key stages to the process of literature searching in systematic reviews were identified. These key stages are consistently reported in the nine guidance documents, suggesting consensus on the key stages of literature searching, and therefore the process of literature searching as a whole, in systematic reviews. Further research to determine the suitability of using the same process of literature searching for all types of systematic review is indicated.
Peer Review reports
Systematic literature searching is recognised as a critical component of the systematic review process. It involves a systematic search for studies and aims for a transparent report of study identification, leaving review stakeholders clear about what was done to identify studies, and how the findings of the review are situated in the relevant evidence.
Information specialists and review teams appear to work from a shared and tacit model of the literature search process. How this tacit model has developed and evolved is unclear, and it has not been explicitly examined before. This is in contrast to the information science literature, which has developed information processing models as an explicit basis for dialogue and empirical testing. Without an explicit model, research in the process of systematic literature searching will remain immature and potentially uneven, and the development of shared information models will be assumed but never articulated.
One way of developing such a conceptual model is by formally examining the implicit “programme theory” as embodied in key methodological texts. The aim of this review is therefore to determine if a shared model of the literature searching process in systematic reviews can be detected across guidance documents and, if so, how this process is reported and supported.
Identifying guidance
Key texts (henceforth referred to as “guidance”) were identified based upon their accessibility to, and prominence within, United Kingdom systematic reviewing practice. The United Kingdom occupies a prominent position in the science of health information retrieval, as quantified by such objective measures as the authorship of papers, the number of Cochrane groups based in the UK, membership and leadership of groups such as the Cochrane Information Retrieval Methods Group, the HTA-I Information Specialists’ Group and historic association with such centres as the UK Cochrane Centre, the NHS Centre for Reviews and Dissemination, the Centre for Evidence Based Medicine and the National Institute for Clinical Excellence (NICE). Coupled with the linguistic dominance of English within medical and health science and the science of systematic reviews more generally, this offers a justification for a purposive sample that favours UK, European and Australian guidance documents.
Nine guidance documents were identified. These documents provide guidance for different types of reviews, namely: reviews of interventions, reviews of health technologies, reviews of qualitative research studies, reviews of social science topics, and reviews to inform guidance.
Whilst these guidance documents occasionally offer additional guidance on other types of systematic reviews, we have focused on the core and stated aims of these documents as they relate to literature searching. Table 1 sets out: the guidance document, the version audited, their core stated focus, and a bibliographical pointer to the main guidance relating to literature searching.
Once a list of key guidance documents was determined, it was checked by six senior information professionals based in the UK for relevance to current literature searching in systematic reviews.
Identifying supporting studies
In addition to identifying guidance, the authors sought to populate an evidence base of supporting studies (henceforth referred to as “studies”) that contribute to existing search practice. Studies were first identified by the authors from their knowledge on this topic area and, subsequently, through systematic citation chasing key studies (‘pearls’ [ 1 ]) located within each key stage of the search process. These studies are identified in Additional file 1 : Appendix Table 1. Citation chasing was conducted by analysing the bibliography of references for each study (backwards citation chasing) and through Google Scholar (forward citation chasing). A search of PubMed using the systematic review methods filter was undertaken in August 2017 (see Additional file 1 ). The search terms used were: (literature search*[Title/Abstract]) AND sysrev_methods[sb] and 586 results were returned. These results were sifted for relevance to the key stages in Fig. 1 by CC.
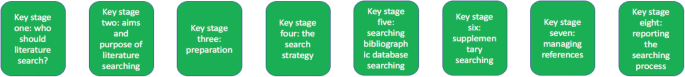
The key stages of literature search guidance as identified from nine key texts
Extracting the data
To reveal the implicit process of literature searching within each guidance document, the relevant sections (chapters) on literature searching were read and re-read, with the aim of determining key methodological stages. We defined a key methodological stage as a distinct step in the overall process for which specific guidance is reported, and action is taken, that collectively would result in a completed literature search.
The chapter or section sub-heading for each methodological stage was extracted into a table using the exact language as reported in each guidance document. The lead author (CC) then read and re-read these data, and the paragraphs of the document to which the headings referred, summarising section details. This table was then reviewed, using comparison and contrast to identify agreements and areas of unique guidance. Consensus across multiple guidelines was used to inform selection of ‘key stages’ in the process of literature searching.
Having determined the key stages to literature searching, we then read and re-read the sections relating to literature searching again, extracting specific detail relating to the methodological process of literature searching within each key stage. Again, the guidance was then read and re-read, first on a document-by-document-basis and, secondly, across all the documents above, to identify both commonalities and areas of unique guidance.
Results and discussion
Our findings.
We were able to identify consensus across the guidance on literature searching for systematic reviews suggesting a shared implicit model within the information retrieval community. Whilst the structure of the guidance varies between documents, the same key stages are reported, even where the core focus of each document is different. We were able to identify specific areas of unique guidance, where a document reported guidance not summarised in other documents, together with areas of consensus across guidance.
Unique guidance
Only one document provided guidance on the topic of when to stop searching [ 2 ]. This guidance from 2005 anticipates a topic of increasing importance with the current interest in time-limited (i.e. “rapid”) reviews. Quality assurance (or peer review) of literature searches was only covered in two guidance documents [ 3 , 4 ]. This topic has emerged as increasingly important as indicated by the development of the PRESS instrument [ 5 ]. Text mining was discussed in four guidance documents [ 4 , 6 , 7 , 8 ] where the automation of some manual review work may offer efficiencies in literature searching [ 8 ].
Agreement between guidance: Defining the key stages of literature searching
Where there was agreement on the process, we determined that this constituted a key stage in the process of literature searching to inform systematic reviews.
From the guidance, we determined eight key stages that relate specifically to literature searching in systematic reviews. These are summarised at Fig. 1 . The data extraction table to inform Fig. 1 is reported in Table 2 . Table 2 reports the areas of common agreement and it demonstrates that the language used to describe key stages and processes varies significantly between guidance documents.
For each key stage, we set out the specific guidance, followed by discussion on how this guidance is situated within the wider literature.
Key stage one: Deciding who should undertake the literature search
The guidance.
Eight documents provided guidance on who should undertake literature searching in systematic reviews [ 2 , 4 , 6 , 7 , 8 , 9 , 10 , 11 ]. The guidance affirms that people with relevant expertise of literature searching should ‘ideally’ be included within the review team [ 6 ]. Information specialists (or information scientists), librarians or trial search co-ordinators (TSCs) are indicated as appropriate researchers in six guidance documents [ 2 , 7 , 8 , 9 , 10 , 11 ].
How the guidance corresponds to the published studies
The guidance is consistent with studies that call for the involvement of information specialists and librarians in systematic reviews [ 12 , 13 , 14 , 15 , 16 , 17 , 18 , 19 , 20 , 21 , 22 , 23 , 24 , 25 , 26 ] and which demonstrate how their training as ‘expert searchers’ and ‘analysers and organisers of data’ can be put to good use [ 13 ] in a variety of roles [ 12 , 16 , 20 , 21 , 24 , 25 , 26 ]. These arguments make sense in the context of the aims and purposes of literature searching in systematic reviews, explored below. The need for ‘thorough’ and ‘replicable’ literature searches was fundamental to the guidance and recurs in key stage two. Studies have found poor reporting, and a lack of replicable literature searches, to be a weakness in systematic reviews [ 17 , 18 , 27 , 28 ] and they argue that involvement of information specialists/ librarians would be associated with better reporting and better quality literature searching. Indeed, Meert et al. [ 29 ] demonstrated that involving a librarian as a co-author to a systematic review correlated with a higher score in the literature searching component of a systematic review [ 29 ]. As ‘new styles’ of rapid and scoping reviews emerge, where decisions on how to search are more iterative and creative, a clear role is made here too [ 30 ].
Knowing where to search for studies was noted as important in the guidance, with no agreement as to the appropriate number of databases to be searched [ 2 , 6 ]. Database (and resource selection more broadly) is acknowledged as a relevant key skill of information specialists and librarians [ 12 , 15 , 16 , 31 ].
Whilst arguments for including information specialists and librarians in the process of systematic review might be considered self-evident, Koffel and Rethlefsen [ 31 ] have questioned if the necessary involvement is actually happening [ 31 ].
Key stage two: Determining the aim and purpose of a literature search
The aim: Five of the nine guidance documents use adjectives such as ‘thorough’, ‘comprehensive’, ‘transparent’ and ‘reproducible’ to define the aim of literature searching [ 6 , 7 , 8 , 9 , 10 ]. Analogous phrases were present in a further three guidance documents, namely: ‘to identify the best available evidence’ [ 4 ] or ‘the aim of the literature search is not to retrieve everything. It is to retrieve everything of relevance’ [ 2 ] or ‘A systematic literature search aims to identify all publications relevant to the particular research question’ [ 3 ]. The Joanna Briggs Institute reviewers’ manual was the only guidance document where a clear statement on the aim of literature searching could not be identified. The purpose of literature searching was defined in three guidance documents, namely to minimise bias in the resultant review [ 6 , 8 , 10 ]. Accordingly, eight of nine documents clearly asserted that thorough and comprehensive literature searches are required as a potential mechanism for minimising bias.
The need for thorough and comprehensive literature searches appears as uniform within the eight guidance documents that describe approaches to literature searching in systematic reviews of effectiveness. Reviews of effectiveness (of intervention or cost), accuracy and prognosis, require thorough and comprehensive literature searches to transparently produce a reliable estimate of intervention effect. The belief that all relevant studies have been ‘comprehensively’ identified, and that this process has been ‘transparently’ reported, increases confidence in the estimate of effect and the conclusions that can be drawn [ 32 ]. The supporting literature exploring the need for comprehensive literature searches focuses almost exclusively on reviews of intervention effectiveness and meta-analysis. Different ‘styles’ of review may have different standards however; the alternative, offered by purposive sampling, has been suggested in the specific context of qualitative evidence syntheses [ 33 ].
What is a comprehensive literature search?
Whilst the guidance calls for thorough and comprehensive literature searches, it lacks clarity on what constitutes a thorough and comprehensive literature search, beyond the implication that all of the literature search methods in Table 2 should be used to identify studies. Egger et al. [ 34 ], in an empirical study evaluating the importance of comprehensive literature searches for trials in systematic reviews, defined a comprehensive search for trials as:
a search not restricted to English language;
where Cochrane CENTRAL or at least two other electronic databases had been searched (such as MEDLINE or EMBASE); and
at least one of the following search methods has been used to identify unpublished trials: searches for (I) conference abstracts, (ii) theses, (iii) trials registers; and (iv) contacts with experts in the field [ 34 ].
Tricco et al. (2008) used a similar threshold of bibliographic database searching AND a supplementary search method in a review when examining the risk of bias in systematic reviews. Their criteria were: one database (limited using the Cochrane Highly Sensitive Search Strategy (HSSS)) and handsearching [ 35 ].
Together with the guidance, this would suggest that comprehensive literature searching requires the use of BOTH bibliographic database searching AND supplementary search methods.
Comprehensiveness in literature searching, in the sense of how much searching should be undertaken, remains unclear. Egger et al. recommend that ‘investigators should consider the type of literature search and degree of comprehension that is appropriate for the review in question, taking into account budget and time constraints’ [ 34 ]. This view tallies with the Cochrane Handbook, which stipulates clearly, that study identification should be undertaken ‘within resource limits’ [ 9 ]. This would suggest that the limitations to comprehension are recognised but it raises questions on how this is decided and reported [ 36 ].
What is the point of comprehensive literature searching?
The purpose of thorough and comprehensive literature searches is to avoid missing key studies and to minimize bias [ 6 , 8 , 10 , 34 , 37 , 38 , 39 ] since a systematic review based only on published (or easily accessible) studies may have an exaggerated effect size [ 35 ]. Felson (1992) sets out potential biases that could affect the estimate of effect in a meta-analysis [ 40 ] and Tricco et al. summarize the evidence concerning bias and confounding in systematic reviews [ 35 ]. Egger et al. point to non-publication of studies, publication bias, language bias and MEDLINE bias, as key biases [ 34 , 35 , 40 , 41 , 42 , 43 , 44 , 45 , 46 ]. Comprehensive searches are not the sole factor to mitigate these biases but their contribution is thought to be significant [ 2 , 32 , 34 ]. Fehrmann (2011) suggests that ‘the search process being described in detail’ and that, where standard comprehensive search techniques have been applied, increases confidence in the search results [ 32 ].
Does comprehensive literature searching work?
Egger et al., and other study authors, have demonstrated a change in the estimate of intervention effectiveness where relevant studies were excluded from meta-analysis [ 34 , 47 ]. This would suggest that missing studies in literature searching alters the reliability of effectiveness estimates. This is an argument for comprehensive literature searching. Conversely, Egger et al. found that ‘comprehensive’ searches still missed studies and that comprehensive searches could, in fact, introduce bias into a review rather than preventing it, through the identification of low quality studies then being included in the meta-analysis [ 34 ]. Studies query if identifying and including low quality or grey literature studies changes the estimate of effect [ 43 , 48 ] and question if time is better invested updating systematic reviews rather than searching for unpublished studies [ 49 ], or mapping studies for review as opposed to aiming for high sensitivity in literature searching [ 50 ].
Aim and purpose beyond reviews of effectiveness
The need for comprehensive literature searches is less certain in reviews of qualitative studies, and for reviews where a comprehensive identification of studies is difficult to achieve (for example, in Public health) [ 33 , 51 , 52 , 53 , 54 , 55 ]. Literature searching for qualitative studies, and in public health topics, typically generates a greater number of studies to sift than in reviews of effectiveness [ 39 ] and demonstrating the ‘value’ of studies identified or missed is harder [ 56 ], since the study data do not typically support meta-analysis. Nussbaumer-Streit et al. (2016) have registered a review protocol to assess whether abbreviated literature searches (as opposed to comprehensive literature searches) has an impact on conclusions across multiple bodies of evidence, not only on effect estimates [ 57 ] which may develop this understanding. It may be that decision makers and users of systematic reviews are willing to trade the certainty from a comprehensive literature search and systematic review in exchange for different approaches to evidence synthesis [ 58 ], and that comprehensive literature searches are not necessarily a marker of literature search quality, as previously thought [ 36 ]. Different approaches to literature searching [ 37 , 38 , 59 , 60 , 61 , 62 ] and developing the concept of when to stop searching are important areas for further study [ 36 , 59 ].
The study by Nussbaumer-Streit et al. has been published since the submission of this literature review [ 63 ]. Nussbaumer-Streit et al. (2018) conclude that abbreviated literature searches are viable options for rapid evidence syntheses, if decision-makers are willing to trade the certainty from a comprehensive literature search and systematic review, but that decision-making which demands detailed scrutiny should still be based on comprehensive literature searches [ 63 ].
Key stage three: Preparing for the literature search
Six documents provided guidance on preparing for a literature search [ 2 , 3 , 6 , 7 , 9 , 10 ]. The Cochrane Handbook clearly stated that Cochrane authors (i.e. researchers) should seek advice from a trial search co-ordinator (i.e. a person with specific skills in literature searching) ‘before’ starting a literature search [ 9 ].
Two key tasks were perceptible in preparing for a literature searching [ 2 , 6 , 7 , 10 , 11 ]. First, to determine if there are any existing or on-going reviews, or if a new review is justified [ 6 , 11 ]; and, secondly, to develop an initial literature search strategy to estimate the volume of relevant literature (and quality of a small sample of relevant studies [ 10 ]) and indicate the resources required for literature searching and the review of the studies that follows [ 7 , 10 ].
Three documents summarised guidance on where to search to determine if a new review was justified [ 2 , 6 , 11 ]. These focused on searching databases of systematic reviews (The Cochrane Database of Systematic Reviews (CDSR) and the Database of Abstracts of Reviews of Effects (DARE)), institutional registries (including PROSPERO), and MEDLINE [ 6 , 11 ]. It is worth noting, however, that as of 2015, DARE (and NHS EEDs) are no longer being updated and so the relevance of this (these) resource(s) will diminish over-time [ 64 ]. One guidance document, ‘Systematic reviews in the Social Sciences’, noted, however, that databases are not the only source of information and unpublished reports, conference proceeding and grey literature may also be required, depending on the nature of the review question [ 2 ].
Two documents reported clearly that this preparation (or ‘scoping’) exercise should be undertaken before the actual search strategy is developed [ 7 , 10 ]).
The guidance offers the best available source on preparing the literature search with the published studies not typically reporting how their scoping informed the development of their search strategies nor how their search approaches were developed. Text mining has been proposed as a technique to develop search strategies in the scoping stages of a review although this work is still exploratory [ 65 ]. ‘Clustering documents’ and word frequency analysis have also been tested to identify search terms and studies for review [ 66 , 67 ]. Preparing for literature searches and scoping constitutes an area for future research.
Key stage four: Designing the search strategy
The Population, Intervention, Comparator, Outcome (PICO) structure was the commonly reported structure promoted to design a literature search strategy. Five documents suggested that the eligibility criteria or review question will determine which concepts of PICO will be populated to develop the search strategy [ 1 , 4 , 7 , 8 , 9 ]. The NICE handbook promoted multiple structures, namely PICO, SPICE (Setting, Perspective, Intervention, Comparison, Evaluation) and multi-stranded approaches [ 4 ].
With the exclusion of The Joanna Briggs Institute reviewers’ manual, the guidance offered detail on selecting key search terms, synonyms, Boolean language, selecting database indexing terms and combining search terms. The CEE handbook suggested that ‘search terms may be compiled with the help of the commissioning organisation and stakeholders’ [ 10 ].
The use of limits, such as language or date limits, were discussed in all documents [ 2 , 3 , 4 , 6 , 7 , 8 , 9 , 10 , 11 ].
Search strategy structure
The guidance typically relates to reviews of intervention effectiveness so PICO – with its focus on intervention and comparator - is the dominant model used to structure literature search strategies [ 68 ]. PICOs – where the S denotes study design - is also commonly used in effectiveness reviews [ 6 , 68 ]. As the NICE handbook notes, alternative models to structure literature search strategies have been developed and tested. Booth provides an overview on formulating questions for evidence based practice [ 69 ] and has developed a number of alternatives to the PICO structure, namely: BeHEMoTh (Behaviour of interest; Health context; Exclusions; Models or Theories) for use when systematically identifying theory [ 55 ]; SPICE (Setting, Perspective, Intervention, Comparison, Evaluation) for identification of social science and evaluation studies [ 69 ] and, working with Cooke and colleagues, SPIDER (Sample, Phenomenon of Interest, Design, Evaluation, Research type) [ 70 ]. SPIDER has been compared to PICO and PICOs in a study by Methley et al. [ 68 ].
The NICE handbook also suggests the use of multi-stranded approaches to developing literature search strategies [ 4 ]. Glanville developed this idea in a study by Whitting et al. [ 71 ] and a worked example of this approach is included in the development of a search filter by Cooper et al. [ 72 ].
Writing search strategies: Conceptual and objective approaches
Hausner et al. [ 73 ] provide guidance on writing literature search strategies, delineating between conceptually and objectively derived approaches. The conceptual approach, advocated by and explained in the guidance documents, relies on the expertise of the literature searcher to identify key search terms and then develop key terms to include synonyms and controlled syntax. Hausner and colleagues set out the objective approach [ 73 ] and describe what may be done to validate it [ 74 ].
The use of limits
The guidance documents offer direction on the use of limits within a literature search. Limits can be used to focus literature searching to specific study designs or by other markers (such as by date) which limits the number of studies returned by a literature search. The use of limits should be described and the implications explored [ 34 ] since limiting literature searching can introduce bias (explored above). Craven et al. have suggested the use of a supporting narrative to explain decisions made in the process of developing literature searches and this advice would usefully capture decisions on the use of search limits [ 75 ].
Key stage five: Determining the process of literature searching and deciding where to search (bibliographic database searching)
Table 2 summarises the process of literature searching as reported in each guidance document. Searching bibliographic databases was consistently reported as the ‘first step’ to literature searching in all nine guidance documents.
Three documents reported specific guidance on where to search, in each case specific to the type of review their guidance informed, and as a minimum requirement [ 4 , 9 , 11 ]. Seven of the key guidance documents suggest that the selection of bibliographic databases depends on the topic of review [ 2 , 3 , 4 , 6 , 7 , 8 , 10 ], with two documents noting the absence of an agreed standard on what constitutes an acceptable number of databases searched [ 2 , 6 ].
The guidance documents summarise ‘how to’ search bibliographic databases in detail and this guidance is further contextualised above in terms of developing the search strategy. The documents provide guidance of selecting bibliographic databases, in some cases stating acceptable minima (i.e. The Cochrane Handbook states Cochrane CENTRAL, MEDLINE and EMBASE), and in other cases simply listing bibliographic database available to search. Studies have explored the value in searching specific bibliographic databases, with Wright et al. (2015) noting the contribution of CINAHL in identifying qualitative studies [ 76 ], Beckles et al. (2013) questioning the contribution of CINAHL to identifying clinical studies for guideline development [ 77 ], and Cooper et al. (2015) exploring the role of UK-focused bibliographic databases to identify UK-relevant studies [ 78 ]. The host of the database (e.g. OVID or ProQuest) has been shown to alter the search returns offered. Younger and Boddy [ 79 ] report differing search returns from the same database (AMED) but where the ‘host’ was different [ 79 ].
The average number of bibliographic database searched in systematic reviews has risen in the period 1994–2014 (from 1 to 4) [ 80 ] but there remains (as attested to by the guidance) no consensus on what constitutes an acceptable number of databases searched [ 48 ]. This is perhaps because thinking about the number of databases searched is the wrong question, researchers should be focused on which databases were searched and why, and which databases were not searched and why. The discussion should re-orientate to the differential value of sources but researchers need to think about how to report this in studies to allow findings to be generalised. Bethel (2017) has proposed ‘search summaries’, completed by the literature searcher, to record where included studies were identified, whether from database (and which databases specifically) or supplementary search methods [ 81 ]. Search summaries document both yield and accuracy of searches, which could prospectively inform resource use and decisions to search or not to search specific databases in topic areas. The prospective use of such data presupposes, however, that past searches are a potential predictor of future search performance (i.e. that each topic is to be considered representative and not unique). In offering a body of practice, this data would be of greater practicable use than current studies which are considered as little more than individual case studies [ 82 , 83 , 84 , 85 , 86 , 87 , 88 , 89 , 90 ].
When to database search is another question posed in the literature. Beyer et al. [ 91 ] report that databases can be prioritised for literature searching which, whilst not addressing the question of which databases to search, may at least bring clarity as to which databases to search first [ 91 ]. Paradoxically, this links to studies that suggest PubMed should be searched in addition to MEDLINE (OVID interface) since this improves the currency of systematic reviews [ 92 , 93 ]. Cooper et al. (2017) have tested the idea of database searching not as a primary search method (as suggested in the guidance) but as a supplementary search method in order to manage the volume of studies identified for an environmental effectiveness systematic review. Their case study compared the effectiveness of database searching versus a protocol using supplementary search methods and found that the latter identified more relevant studies for review than searching bibliographic databases [ 94 ].
Key stage six: Determining the process of literature searching and deciding where to search (supplementary search methods)
Table 2 also summaries the process of literature searching which follows bibliographic database searching. As Table 2 sets out, guidance that supplementary literature search methods should be used in systematic reviews recurs across documents, but the order in which these methods are used, and the extent to which they are used, varies. We noted inconsistency in the labelling of supplementary search methods between guidance documents.
Rather than focus on the guidance on how to use the methods (which has been summarised in a recent review [ 95 ]), we focus on the aim or purpose of supplementary search methods.
The Cochrane Handbook reported that ‘efforts’ to identify unpublished studies should be made [ 9 ]. Four guidance documents [ 2 , 3 , 6 , 9 ] acknowledged that searching beyond bibliographic databases was necessary since ‘databases are not the only source of literature’ [ 2 ]. Only one document reported any guidance on determining when to use supplementary methods. The IQWiG handbook reported that the use of handsearching (in their example) could be determined on a ‘case-by-case basis’ which implies that the use of these methods is optional rather than mandatory. This is in contrast to the guidance (above) on bibliographic database searching.
The issue for supplementary search methods is similar in many ways to the issue of searching bibliographic databases: demonstrating value. The purpose and contribution of supplementary search methods in systematic reviews is increasingly acknowledged [ 37 , 61 , 62 , 96 , 97 , 98 , 99 , 100 , 101 ] but understanding the value of the search methods to identify studies and data is unclear. In a recently published review, Cooper et al. (2017) reviewed the literature on supplementary search methods looking to determine the advantages, disadvantages and resource implications of using supplementary search methods [ 95 ]. This review also summarises the key guidance and empirical studies and seeks to address the question on when to use these search methods and when not to [ 95 ]. The guidance is limited in this regard and, as Table 2 demonstrates, offers conflicting advice on the order of searching, and the extent to which these search methods should be used in systematic reviews.
Key stage seven: Managing the references
Five of the documents provided guidance on managing references, for example downloading, de-duplicating and managing the output of literature searches [ 2 , 4 , 6 , 8 , 10 ]. This guidance typically itemised available bibliographic management tools rather than offering guidance on how to use them specifically [ 2 , 4 , 6 , 8 ]. The CEE handbook provided guidance on importing data where no direct export option is available (e.g. web-searching) [ 10 ].
The literature on using bibliographic management tools is not large relative to the number of ‘how to’ videos on platforms such as YouTube (see for example [ 102 ]). These YouTube videos confirm the overall lack of ‘how to’ guidance identified in this study and offer useful instruction on managing references. Bramer et al. set out methods for de-duplicating data and reviewing references in Endnote [ 103 , 104 ] and Gall tests the direct search function within Endnote to access databases such as PubMed, finding a number of limitations [ 105 ]. Coar et al. and Ahmed et al. consider the role of the free-source tool, Zotero [ 106 , 107 ]. Managing references is a key administrative function in the process of review particularly for documenting searches in PRISMA guidance.
Key stage eight: Documenting the search
The Cochrane Handbook was the only guidance document to recommend a specific reporting guideline: Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [ 9 ]. Six documents provided guidance on reporting the process of literature searching with specific criteria to report [ 3 , 4 , 6 , 8 , 9 , 10 ]. There was consensus on reporting: the databases searched (and the host searched by), the search strategies used, and any use of limits (e.g. date, language, search filters (The CRD handbook called for these limits to be justified [ 6 ])). Three guidance documents reported that the number of studies identified should be recorded [ 3 , 6 , 10 ]. The number of duplicates identified [ 10 ], the screening decisions [ 3 ], a comprehensive list of grey literature sources searched (and full detail for other supplementary search methods) [ 8 ], and an annotation of search terms tested but not used [ 4 ] were identified as unique items in four documents.
The Cochrane Handbook was the only guidance document to note that the full search strategies for each database should be included in the Additional file 1 of the review [ 9 ].
All guidance documents should ultimately deliver completed systematic reviews that fulfil the requirements of the PRISMA reporting guidelines [ 108 ]. The guidance broadly requires the reporting of data that corresponds with the requirements of the PRISMA statement although documents typically ask for diverse and additional items [ 108 ]. In 2008, Sampson et al. observed a lack of consensus on reporting search methods in systematic reviews [ 109 ] and this remains the case as of 2017, as evidenced in the guidance documents, and in spite of the publication of the PRISMA guidelines in 2009 [ 110 ]. It is unclear why the collective guidance does not more explicitly endorse adherence to the PRISMA guidance.
Reporting of literature searching is a key area in systematic reviews since it sets out clearly what was done and how the conclusions of the review can be believed [ 52 , 109 ]. Despite strong endorsement in the guidance documents, specifically supported in PRISMA guidance, and other related reporting standards too (such as ENTREQ for qualitative evidence synthesis, STROBE for reviews of observational studies), authors still highlight the prevalence of poor standards of literature search reporting [ 31 , 110 , 111 , 112 , 113 , 114 , 115 , 116 , 117 , 118 , 119 ]. To explore issues experienced by authors in reporting literature searches, and look at uptake of PRISMA, Radar et al. [ 120 ] surveyed over 260 review authors to determine common problems and their work summaries the practical aspects of reporting literature searching [ 120 ]. Atkinson et al. [ 121 ] have also analysed reporting standards for literature searching, summarising recommendations and gaps for reporting search strategies [ 121 ].
One area that is less well covered by the guidance, but nevertheless appears in this literature, is the quality appraisal or peer review of literature search strategies. The PRESS checklist is the most prominent and it aims to develop evidence-based guidelines to peer review of electronic search strategies [ 5 , 122 , 123 ]. A corresponding guideline for documentation of supplementary search methods does not yet exist although this idea is currently being explored.
How the reporting of the literature searching process corresponds to critical appraisal tools is an area for further research. In the survey undertaken by Radar et al. (2014), 86% of survey respondents (153/178) identified a need for further guidance on what aspects of the literature search process to report [ 120 ]. The PRISMA statement offers a brief summary of what to report but little practical guidance on how to report it [ 108 ]. Critical appraisal tools for systematic reviews, such as AMSTAR 2 (Shea et al. [ 124 ]) and ROBIS (Whiting et al. [ 125 ]), can usefully be read alongside PRISMA guidance, since they offer greater detail on how the reporting of the literature search will be appraised and, therefore, they offer a proxy on what to report [ 124 , 125 ]. Further research in the form of a study which undertakes a comparison between PRISMA and quality appraisal checklists for systematic reviews would seem to begin addressing the call, identified by Radar et al., for further guidance on what to report [ 120 ].
Limitations
Other handbooks exist.
A potential limitation of this literature review is the focus on guidance produced in Europe (the UK specifically) and Australia. We justify the decision for our selection of the nine guidance documents reviewed in this literature review in section “ Identifying guidance ”. In brief, these nine guidance documents were selected as the most relevant health care guidance that inform UK systematic reviewing practice, given that the UK occupies a prominent position in the science of health information retrieval. We acknowledge the existence of other guidance documents, such as those from North America (e.g. the Agency for Healthcare Research and Quality (AHRQ) [ 126 ], The Institute of Medicine [ 127 ] and the guidance and resources produced by the Canadian Agency for Drugs and Technologies in Health (CADTH) [ 128 ]). We comment further on this directly below.
The handbooks are potentially linked to one another
What is not clear is the extent to which the guidance documents inter-relate or provide guidance uniquely. The Cochrane Handbook, first published in 1994, is notably a key source of reference in guidance and systematic reviews beyond Cochrane reviews. It is not clear to what extent broadening the sample of guidance handbooks to include North American handbooks, and guidance handbooks from other relevant countries too, would alter the findings of this literature review or develop further support for the process model. Since we cannot be clear, we raise this as a potential limitation of this literature review. On our initial review of a sample of North American, and other, guidance documents (before selecting the guidance documents considered in this review), however, we do not consider that the inclusion of these further handbooks would alter significantly the findings of this literature review.
This is a literature review
A further limitation of this review was that the review of published studies is not a systematic review of the evidence for each key stage. It is possible that other relevant studies could help contribute to the exploration and development of the key stages identified in this review.
This literature review would appear to demonstrate the existence of a shared model of the literature searching process in systematic reviews. We call this model ‘the conventional approach’, since it appears to be common convention in nine different guidance documents.
The findings reported above reveal eight key stages in the process of literature searching for systematic reviews. These key stages are consistently reported in the nine guidance documents which suggests consensus on the key stages of literature searching, and therefore the process of literature searching as a whole, in systematic reviews.
In Table 2 , we demonstrate consensus regarding the application of literature search methods. All guidance documents distinguish between primary and supplementary search methods. Bibliographic database searching is consistently the first method of literature searching referenced in each guidance document. Whilst the guidance uniformly supports the use of supplementary search methods, there is little evidence for a consistent process with diverse guidance across documents. This may reflect differences in the core focus across each document, linked to differences in identifying effectiveness studies or qualitative studies, for instance.
Eight of the nine guidance documents reported on the aims of literature searching. The shared understanding was that literature searching should be thorough and comprehensive in its aim and that this process should be reported transparently so that that it could be reproduced. Whilst only three documents explicitly link this understanding to minimising bias, it is clear that comprehensive literature searching is implicitly linked to ‘not missing relevant studies’ which is approximately the same point.
Defining the key stages in this review helps categorise the scholarship available, and it prioritises areas for development or further study. The supporting studies on preparing for literature searching (key stage three, ‘preparation’) were, for example, comparatively few, and yet this key stage represents a decisive moment in literature searching for systematic reviews. It is where search strategy structure is determined, search terms are chosen or discarded, and the resources to be searched are selected. Information specialists, librarians and researchers, are well placed to develop these and other areas within the key stages we identify.
This review calls for further research to determine the suitability of using the conventional approach. The publication dates of the guidance documents which underpin the conventional approach may raise questions as to whether the process which they each report remains valid for current systematic literature searching. In addition, it may be useful to test whether it is desirable to use the same process model of literature searching for qualitative evidence synthesis as that for reviews of intervention effectiveness, which this literature review demonstrates is presently recommended best practice.
Abbreviations
Behaviour of interest; Health context; Exclusions; Models or Theories
Cochrane Database of Systematic Reviews
The Cochrane Central Register of Controlled Trials
Database of Abstracts of Reviews of Effects
Enhancing transparency in reporting the synthesis of qualitative research
Institute for Quality and Efficiency in Healthcare
National Institute for Clinical Excellence
Population, Intervention, Comparator, Outcome
Preferred Reporting Items for Systematic Reviews and Meta-Analyses
Setting, Perspective, Intervention, Comparison, Evaluation
Sample, Phenomenon of Interest, Design, Evaluation, Research type
STrengthening the Reporting of OBservational studies in Epidemiology
Trial Search Co-ordinators
Booth A. Unpacking your literature search toolbox: on search styles and tactics. Health Information & Libraries Journal. 2008;25(4):313–7.
Article Google Scholar
Petticrew M, Roberts H. Systematic reviews in the social sciences: a practical guide. Oxford: Blackwell Publishing Ltd; 2006.
Book Google Scholar
Institute for Quality and Efficiency in Health Care (IQWiG). IQWiG Methods Resources. 7 Information retrieval 2014 [Available from: https://www.ncbi.nlm.nih.gov/books/NBK385787/ .
NICE: National Institute for Health and Care Excellence. Developing NICE guidelines: the manual 2014. Available from: https://www.nice.org.uk/media/default/about/what-we-do/our-programmes/developing-nice-guidelines-the-manual.pdf .
Sampson M. MJ, Lefebvre C, Moher D, Grimshaw J. Peer Review of Electronic Search Strategies: PRESS; 2008.
Google Scholar
Centre for Reviews & Dissemination. Systematic reviews – CRD’s guidance for undertaking reviews in healthcare. York: Centre for Reviews and Dissemination, University of York; 2009.
eunetha: European Network for Health Technology Assesment Process of information retrieval for systematic reviews and health technology assessments on clinical effectiveness 2016. Available from: http://www.eunethta.eu/sites/default/files/Guideline_Information_Retrieval_V1-1.pdf .
Kugley SWA, Thomas J, Mahood Q, Jørgensen AMK, Hammerstrøm K, Sathe N. Searching for studies: a guide to information retrieval for Campbell systematic reviews. Oslo: Campbell Collaboration. 2017; Available from: https://www.campbellcollaboration.org/library/searching-for-studies-information-retrieval-guide-campbell-reviews.html
Lefebvre C, Manheimer E, Glanville J. Chapter 6: searching for studies. In: JPT H, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions; 2011.
Collaboration for Environmental Evidence. Guidelines for Systematic Review and Evidence Synthesis in Environmental Management.: Environmental Evidence:; 2013. Available from: http://www.environmentalevidence.org/wp-content/uploads/2017/01/Review-guidelines-version-4.2-final-update.pdf .
The Joanna Briggs Institute. Joanna Briggs institute reviewers’ manual. 2014th ed: the Joanna Briggs institute; 2014. Available from: https://joannabriggs.org/assets/docs/sumari/ReviewersManual-2014.pdf
Beverley CA, Booth A, Bath PA. The role of the information specialist in the systematic review process: a health information case study. Health Inf Libr J. 2003;20(2):65–74.
Article CAS Google Scholar
Harris MR. The librarian's roles in the systematic review process: a case study. Journal of the Medical Library Association. 2005;93(1):81–7.
PubMed PubMed Central Google Scholar
Egger JB. Use of recommended search strategies in systematic reviews and the impact of librarian involvement: a cross-sectional survey of recent authors. PLoS One. 2015;10(5):e0125931.
Li L, Tian J, Tian H, Moher D, Liang F, Jiang T, et al. Network meta-analyses could be improved by searching more sources and by involving a librarian. J Clin Epidemiol. 2014;67(9):1001–7.
Article PubMed Google Scholar
McGowan J, Sampson M. Systematic reviews need systematic searchers. J Med Libr Assoc. 2005;93(1):74–80.
Rethlefsen ML, Farrell AM, Osterhaus Trzasko LC, Brigham TJ. Librarian co-authors correlated with higher quality reported search strategies in general internal medicine systematic reviews. J Clin Epidemiol. 2015;68(6):617–26.
Weller AC. Mounting evidence that librarians are essential for comprehensive literature searches for meta-analyses and Cochrane reports. J Med Libr Assoc. 2004;92(2):163–4.
Swinkels A, Briddon J, Hall J. Two physiotherapists, one librarian and a systematic literature review: collaboration in action. Health Info Libr J. 2006;23(4):248–56.
Foster M. An overview of the role of librarians in systematic reviews: from expert search to project manager. EAHIL. 2015;11(3):3–7.
Lawson L. OPERATING OUTSIDE LIBRARY WALLS 2004.
Vassar M, Yerokhin V, Sinnett PM, Weiher M, Muckelrath H, Carr B, et al. Database selection in systematic reviews: an insight through clinical neurology. Health Inf Libr J. 2017;34(2):156–64.
Townsend WA, Anderson PF, Ginier EC, MacEachern MP, Saylor KM, Shipman BL, et al. A competency framework for librarians involved in systematic reviews. Journal of the Medical Library Association : JMLA. 2017;105(3):268–75.
Cooper ID, Crum JA. New activities and changing roles of health sciences librarians: a systematic review, 1990-2012. Journal of the Medical Library Association : JMLA. 2013;101(4):268–77.
Crum JA, Cooper ID. Emerging roles for biomedical librarians: a survey of current practice, challenges, and changes. Journal of the Medical Library Association : JMLA. 2013;101(4):278–86.
Dudden RF, Protzko SL. The systematic review team: contributions of the health sciences librarian. Med Ref Serv Q. 2011;30(3):301–15.
Golder S, Loke Y, McIntosh HM. Poor reporting and inadequate searches were apparent in systematic reviews of adverse effects. J Clin Epidemiol. 2008;61(5):440–8.
Maggio LA, Tannery NH, Kanter SL. Reproducibility of literature search reporting in medical education reviews. Academic medicine : journal of the Association of American Medical Colleges. 2011;86(8):1049–54.
Meert D, Torabi N, Costella J. Impact of librarians on reporting of the literature searching component of pediatric systematic reviews. Journal of the Medical Library Association : JMLA. 2016;104(4):267–77.
Morris M, Boruff JT, Gore GC. Scoping reviews: establishing the role of the librarian. Journal of the Medical Library Association : JMLA. 2016;104(4):346–54.
Koffel JB, Rethlefsen ML. Reproducibility of search strategies is poor in systematic reviews published in high-impact pediatrics, cardiology and surgery journals: a cross-sectional study. PLoS One. 2016;11(9):e0163309.
Article PubMed PubMed Central CAS Google Scholar
Fehrmann P, Thomas J. Comprehensive computer searches and reporting in systematic reviews. Research Synthesis Methods. 2011;2(1):15–32.
Booth A. Searching for qualitative research for inclusion in systematic reviews: a structured methodological review. Systematic Reviews. 2016;5(1):74.
Article PubMed PubMed Central Google Scholar
Egger M, Juni P, Bartlett C, Holenstein F, Sterne J. How important are comprehensive literature searches and the assessment of trial quality in systematic reviews? Empirical study. Health technology assessment (Winchester, England). 2003;7(1):1–76.
Tricco AC, Tetzlaff J, Sampson M, Fergusson D, Cogo E, Horsley T, et al. Few systematic reviews exist documenting the extent of bias: a systematic review. J Clin Epidemiol. 2008;61(5):422–34.
Booth A. How much searching is enough? Comprehensive versus optimal retrieval for technology assessments. Int J Technol Assess Health Care. 2010;26(4):431–5.
Papaioannou D, Sutton A, Carroll C, Booth A, Wong R. Literature searching for social science systematic reviews: consideration of a range of search techniques. Health Inf Libr J. 2010;27(2):114–22.
Petticrew M. Time to rethink the systematic review catechism? Moving from ‘what works’ to ‘what happens’. Systematic Reviews. 2015;4(1):36.
Betrán AP, Say L, Gülmezoglu AM, Allen T, Hampson L. Effectiveness of different databases in identifying studies for systematic reviews: experience from the WHO systematic review of maternal morbidity and mortality. BMC Med Res Methodol. 2005;5
Felson DT. Bias in meta-analytic research. J Clin Epidemiol. 1992;45(8):885–92.
Article PubMed CAS Google Scholar
Franco A, Malhotra N, Simonovits G. Publication bias in the social sciences: unlocking the file drawer. Science. 2014;345(6203):1502–5.
Hartling L, Featherstone R, Nuspl M, Shave K, Dryden DM, Vandermeer B. Grey literature in systematic reviews: a cross-sectional study of the contribution of non-English reports, unpublished studies and dissertations to the results of meta-analyses in child-relevant reviews. BMC Med Res Methodol. 2017;17(1):64.
Schmucker CM, Blümle A, Schell LK, Schwarzer G, Oeller P, Cabrera L, et al. Systematic review finds that study data not published in full text articles have unclear impact on meta-analyses results in medical research. PLoS One. 2017;12(4):e0176210.
Egger M, Zellweger-Zahner T, Schneider M, Junker C, Lengeler C, Antes G. Language bias in randomised controlled trials published in English and German. Lancet (London, England). 1997;350(9074):326–9.
Moher D, Pham B, Lawson ML, Klassen TP. The inclusion of reports of randomised trials published in languages other than English in systematic reviews. Health technology assessment (Winchester, England). 2003;7(41):1–90.
Pham B, Klassen TP, Lawson ML, Moher D. Language of publication restrictions in systematic reviews gave different results depending on whether the intervention was conventional or complementary. J Clin Epidemiol. 2005;58(8):769–76.
Mills EJ, Kanters S, Thorlund K, Chaimani A, Veroniki A-A, Ioannidis JPA. The effects of excluding treatments from network meta-analyses: survey. BMJ : British Medical Journal. 2013;347
Hartling L, Featherstone R, Nuspl M, Shave K, Dryden DM, Vandermeer B. The contribution of databases to the results of systematic reviews: a cross-sectional study. BMC Med Res Methodol. 2016;16(1):127.
van Driel ML, De Sutter A, De Maeseneer J, Christiaens T. Searching for unpublished trials in Cochrane reviews may not be worth the effort. J Clin Epidemiol. 2009;62(8):838–44.e3.
Buchberger B, Krabbe L, Lux B, Mattivi JT. Evidence mapping for decision making: feasibility versus accuracy - when to abandon high sensitivity in electronic searches. German medical science : GMS e-journal. 2016;14:Doc09.
Lorenc T, Pearson M, Jamal F, Cooper C, Garside R. The role of systematic reviews of qualitative evidence in evaluating interventions: a case study. Research Synthesis Methods. 2012;3(1):1–10.
Gough D. Weight of evidence: a framework for the appraisal of the quality and relevance of evidence. Res Pap Educ. 2007;22(2):213–28.
Barroso J, Gollop CJ, Sandelowski M, Meynell J, Pearce PF, Collins LJ. The challenges of searching for and retrieving qualitative studies. West J Nurs Res. 2003;25(2):153–78.
Britten N, Garside R, Pope C, Frost J, Cooper C. Asking more of qualitative synthesis: a response to Sally Thorne. Qual Health Res. 2017;27(9):1370–6.
Booth A, Carroll C. Systematic searching for theory to inform systematic reviews: is it feasible? Is it desirable? Health Info Libr J. 2015;32(3):220–35.
Kwon Y, Powelson SE, Wong H, Ghali WA, Conly JM. An assessment of the efficacy of searching in biomedical databases beyond MEDLINE in identifying studies for a systematic review on ward closures as an infection control intervention to control outbreaks. Syst Rev. 2014;3:135.
Nussbaumer-Streit B, Klerings I, Wagner G, Titscher V, Gartlehner G. Assessing the validity of abbreviated literature searches for rapid reviews: protocol of a non-inferiority and meta-epidemiologic study. Systematic Reviews. 2016;5:197.
Wagner G, Nussbaumer-Streit B, Greimel J, Ciapponi A, Gartlehner G. Trading certainty for speed - how much uncertainty are decisionmakers and guideline developers willing to accept when using rapid reviews: an international survey. BMC Med Res Methodol. 2017;17(1):121.
Ogilvie D, Hamilton V, Egan M, Petticrew M. Systematic reviews of health effects of social interventions: 1. Finding the evidence: how far should you go? J Epidemiol Community Health. 2005;59(9):804–8.
Royle P, Milne R. Literature searching for randomized controlled trials used in Cochrane reviews: rapid versus exhaustive searches. Int J Technol Assess Health Care. 2003;19(4):591–603.
Pearson M, Moxham T, Ashton K. Effectiveness of search strategies for qualitative research about barriers and facilitators of program delivery. Eval Health Prof. 2011;34(3):297–308.
Levay P, Raynor M, Tuvey D. The Contributions of MEDLINE, Other Bibliographic Databases and Various Search Techniques to NICE Public Health Guidance. 2015. 2015;10(1):19.
Nussbaumer-Streit B, Klerings I, Wagner G, Heise TL, Dobrescu AI, Armijo-Olivo S, et al. Abbreviated literature searches were viable alternatives to comprehensive searches: a meta-epidemiological study. J Clin Epidemiol. 2018;102:1–11.
Briscoe S, Cooper C, Glanville J, Lefebvre C. The loss of the NHS EED and DARE databases and the effect on evidence synthesis and evaluation. Res Synth Methods. 2017;8(3):256–7.
Stansfield C, O'Mara-Eves A, Thomas J. Text mining for search term development in systematic reviewing: A discussion of some methods and challenges. Research Synthesis Methods.n/a-n/a.
Petrova M, Sutcliffe P, Fulford KW, Dale J. Search terms and a validated brief search filter to retrieve publications on health-related values in Medline: a word frequency analysis study. Journal of the American Medical Informatics Association : JAMIA. 2012;19(3):479–88.
Stansfield C, Thomas J, Kavanagh J. 'Clustering' documents automatically to support scoping reviews of research: a case study. Res Synth Methods. 2013;4(3):230–41.
PubMed Google Scholar
Methley AM, Campbell S, Chew-Graham C, McNally R, Cheraghi-Sohi S. PICO, PICOS and SPIDER: a comparison study of specificity and sensitivity in three search tools for qualitative systematic reviews. BMC Health Serv Res. 2014;14:579.
Andrew B. Clear and present questions: formulating questions for evidence based practice. Library Hi Tech. 2006;24(3):355–68.
Cooke A, Smith D, Booth A. Beyond PICO: the SPIDER tool for qualitative evidence synthesis. Qual Health Res. 2012;22(10):1435–43.
Whiting P, Westwood M, Bojke L, Palmer S, Richardson G, Cooper J, et al. Clinical effectiveness and cost-effectiveness of tests for the diagnosis and investigation of urinary tract infection in children: a systematic review and economic model. Health technology assessment (Winchester, England). 2006;10(36):iii-iv, xi-xiii, 1–154.
Cooper C, Levay P, Lorenc T, Craig GM. A population search filter for hard-to-reach populations increased search efficiency for a systematic review. J Clin Epidemiol. 2014;67(5):554–9.
Hausner E, Waffenschmidt S, Kaiser T, Simon M. Routine development of objectively derived search strategies. Systematic Reviews. 2012;1(1):19.
Hausner E, Guddat C, Hermanns T, Lampert U, Waffenschmidt S. Prospective comparison of search strategies for systematic reviews: an objective approach yielded higher sensitivity than a conceptual one. J Clin Epidemiol. 2016;77:118–24.
Craven J, Levay P. Recording database searches for systematic reviews - what is the value of adding a narrative to peer-review checklists? A case study of nice interventional procedures guidance. Evid Based Libr Inf Pract. 2011;6(4):72–87.
Wright K, Golder S, Lewis-Light K. What value is the CINAHL database when searching for systematic reviews of qualitative studies? Syst Rev. 2015;4:104.
Beckles Z, Glover S, Ashe J, Stockton S, Boynton J, Lai R, et al. Searching CINAHL did not add value to clinical questions posed in NICE guidelines. J Clin Epidemiol. 2013;66(9):1051–7.
Cooper C, Rogers M, Bethel A, Briscoe S, Lowe J. A mapping review of the literature on UK-focused health and social care databases. Health Inf Libr J. 2015;32(1):5–22.
Younger P, Boddy K. When is a search not a search? A comparison of searching the AMED complementary health database via EBSCOhost, OVID and DIALOG. Health Inf Libr J. 2009;26(2):126–35.
Lam MT, McDiarmid M. Increasing number of databases searched in systematic reviews and meta-analyses between 1994 and 2014. Journal of the Medical Library Association : JMLA. 2016;104(4):284–9.
Bethel A, editor Search summary tables for systematic reviews: results and findings. HLC Conference 2017a.
Aagaard T, Lund H, Juhl C. Optimizing literature search in systematic reviews - are MEDLINE, EMBASE and CENTRAL enough for identifying effect studies within the area of musculoskeletal disorders? BMC Med Res Methodol. 2016;16(1):161.
Adams CE, Frederick K. An investigation of the adequacy of MEDLINE searches for randomized controlled trials (RCTs) of the effects of mental health care. Psychol Med. 1994;24(3):741–8.
Kelly L, St Pierre-Hansen N. So many databases, such little clarity: searching the literature for the topic aboriginal. Canadian family physician Medecin de famille canadien. 2008;54(11):1572–3.
Lawrence DW. What is lost when searching only one literature database for articles relevant to injury prevention and safety promotion? Injury Prevention. 2008;14(6):401–4.
Lemeshow AR, Blum RE, Berlin JA, Stoto MA, Colditz GA. Searching one or two databases was insufficient for meta-analysis of observational studies. J Clin Epidemiol. 2005;58(9):867–73.
Sampson M, Barrowman NJ, Moher D, Klassen TP, Pham B, Platt R, et al. Should meta-analysts search Embase in addition to Medline? J Clin Epidemiol. 2003;56(10):943–55.
Stevinson C, Lawlor DA. Searching multiple databases for systematic reviews: added value or diminishing returns? Complementary Therapies in Medicine. 2004;12(4):228–32.
Suarez-Almazor ME, Belseck E, Homik J, Dorgan M, Ramos-Remus C. Identifying clinical trials in the medical literature with electronic databases: MEDLINE alone is not enough. Control Clin Trials. 2000;21(5):476–87.
Taylor B, Wylie E, Dempster M, Donnelly M. Systematically retrieving research: a case study evaluating seven databases. Res Soc Work Pract. 2007;17(6):697–706.
Beyer FR, Wright K. Can we prioritise which databases to search? A case study using a systematic review of frozen shoulder management. Health Info Libr J. 2013;30(1):49–58.
Duffy S, de Kock S, Misso K, Noake C, Ross J, Stirk L. Supplementary searches of PubMed to improve currency of MEDLINE and MEDLINE in-process searches via Ovid. Journal of the Medical Library Association : JMLA. 2016;104(4):309–12.
Katchamart W, Faulkner A, Feldman B, Tomlinson G, Bombardier C. PubMed had a higher sensitivity than Ovid-MEDLINE in the search for systematic reviews. J Clin Epidemiol. 2011;64(7):805–7.
Cooper C, Lovell R, Husk K, Booth A, Garside R. Supplementary search methods were more effective and offered better value than bibliographic database searching: a case study from public health and environmental enhancement (in Press). Research Synthesis Methods. 2017;
Cooper C, Booth, A., Britten, N., Garside, R. A comparison of results of empirical studies of supplementary search techniques and recommendations in review methodology handbooks: A methodological review. (In Press). BMC Systematic Reviews. 2017.
Greenhalgh T, Peacock R. Effectiveness and efficiency of search methods in systematic reviews of complex evidence: audit of primary sources. BMJ (Clinical research ed). 2005;331(7524):1064–5.
Article PubMed Central Google Scholar
Hinde S, Spackman E. Bidirectional citation searching to completion: an exploration of literature searching methods. PharmacoEconomics. 2015;33(1):5–11.
Levay P, Ainsworth N, Kettle R, Morgan A. Identifying evidence for public health guidance: a comparison of citation searching with web of science and Google scholar. Res Synth Methods. 2016;7(1):34–45.
McManus RJ, Wilson S, Delaney BC, Fitzmaurice DA, Hyde CJ, Tobias RS, et al. Review of the usefulness of contacting other experts when conducting a literature search for systematic reviews. BMJ (Clinical research ed). 1998;317(7172):1562–3.
Westphal A, Kriston L, Holzel LP, Harter M, von Wolff A. Efficiency and contribution of strategies for finding randomized controlled trials: a case study from a systematic review on therapeutic interventions of chronic depression. Journal of public health research. 2014;3(2):177.
Matthews EJ, Edwards AG, Barker J, Bloor M, Covey J, Hood K, et al. Efficient literature searching in diffuse topics: lessons from a systematic review of research on communicating risk to patients in primary care. Health Libr Rev. 1999;16(2):112–20.
Bethel A. Endnote Training (YouTube Videos) 2017b [Available from: http://medicine.exeter.ac.uk/esmi/workstreams/informationscience/is_resources,_guidance_&_advice/ .
Bramer WM, Giustini D, de Jonge GB, Holland L, Bekhuis T. De-duplication of database search results for systematic reviews in EndNote. Journal of the Medical Library Association : JMLA. 2016;104(3):240–3.
Bramer WM, Milic J, Mast F. Reviewing retrieved references for inclusion in systematic reviews using EndNote. Journal of the Medical Library Association : JMLA. 2017;105(1):84–7.
Gall C, Brahmi FA. Retrieval comparison of EndNote to search MEDLINE (Ovid and PubMed) versus searching them directly. Medical reference services quarterly. 2004;23(3):25–32.
Ahmed KK, Al Dhubaib BE. Zotero: a bibliographic assistant to researcher. J Pharmacol Pharmacother. 2011;2(4):303–5.
Coar JT, Sewell JP. Zotero: harnessing the power of a personal bibliographic manager. Nurse Educ. 2010;35(5):205–7.
Moher D, Liberati A, Tetzlaff J, Altman DG, The PG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097.
Sampson M, McGowan J, Tetzlaff J, Cogo E, Moher D. No consensus exists on search reporting methods for systematic reviews. J Clin Epidemiol. 2008;61(8):748–54.
Toews LC. Compliance of systematic reviews in veterinary journals with preferred reporting items for systematic reviews and meta-analysis (PRISMA) literature search reporting guidelines. Journal of the Medical Library Association : JMLA. 2017;105(3):233–9.
Booth A. "brimful of STARLITE": toward standards for reporting literature searches. Journal of the Medical Library Association : JMLA. 2006;94(4):421–9. e205
Faggion CM Jr, Wu YC, Tu YK, Wasiak J. Quality of search strategies reported in systematic reviews published in stereotactic radiosurgery. Br J Radiol. 2016;89(1062):20150878.
Mullins MM, DeLuca JB, Crepaz N, Lyles CM. Reporting quality of search methods in systematic reviews of HIV behavioral interventions (2000–2010): are the searches clearly explained, systematic and reproducible? Research Synthesis Methods. 2014;5(2):116–30.
Yoshii A, Plaut DA, McGraw KA, Anderson MJ, Wellik KE. Analysis of the reporting of search strategies in Cochrane systematic reviews. Journal of the Medical Library Association : JMLA. 2009;97(1):21–9.
Bigna JJ, Um LN, Nansseu JR. A comparison of quality of abstracts of systematic reviews including meta-analysis of randomized controlled trials in high-impact general medicine journals before and after the publication of PRISMA extension for abstracts: a systematic review and meta-analysis. Syst Rev. 2016;5(1):174.
Akhigbe T, Zolnourian A, Bulters D. Compliance of systematic reviews articles in brain arteriovenous malformation with PRISMA statement guidelines: review of literature. Journal of clinical neuroscience : official journal of the Neurosurgical Society of Australasia. 2017;39:45–8.
Tao KM, Li XQ, Zhou QH, Moher D, Ling CQ, Yu WF. From QUOROM to PRISMA: a survey of high-impact medical journals' instructions to authors and a review of systematic reviews in anesthesia literature. PLoS One. 2011;6(11):e27611.
Wasiak J, Tyack Z, Ware R. Goodwin N. Jr. Poor methodological quality and reporting standards of systematic reviews in burn care management. International wound journal: Faggion CM; 2016.
Tam WW, Lo KK, Khalechelvam P. Endorsement of PRISMA statement and quality of systematic reviews and meta-analyses published in nursing journals: a cross-sectional study. BMJ Open. 2017;7(2):e013905.
Rader T, Mann M, Stansfield C, Cooper C, Sampson M. Methods for documenting systematic review searches: a discussion of common issues. Res Synth Methods. 2014;5(2):98–115.
Atkinson KM, Koenka AC, Sanchez CE, Moshontz H, Cooper H. Reporting standards for literature searches and report inclusion criteria: making research syntheses more transparent and easy to replicate. Res Synth Methods. 2015;6(1):87–95.
McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS peer review of electronic search strategies: 2015 guideline statement. J Clin Epidemiol. 2016;75:40–6.
Sampson M, McGowan J, Cogo E, Grimshaw J, Moher D, Lefebvre C. An evidence-based practice guideline for the peer review of electronic search strategies. J Clin Epidemiol. 2009;62(9):944–52.
Shea BJ, Reeves BC, Wells G, Thuku M, Hamel C, Moran J, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ (Clinical research ed). 2017;358.
Whiting P, Savović J, Higgins JPT, Caldwell DM, Reeves BC, Shea B, et al. ROBIS: a new tool to assess risk of bias in systematic reviews was developed. J Clin Epidemiol. 2016;69:225–34.
Relevo R, Balshem H. Finding evidence for comparing medical interventions: AHRQ and the effective health care program. J Clin Epidemiol. 2011;64(11):1168–77.
Medicine Io. Standards for Systematic Reviews 2011 [Available from: http://www.nationalacademies.org/hmd/Reports/2011/Finding-What-Works-in-Health-Care-Standards-for-Systematic-Reviews/Standards.aspx .
CADTH: Resources 2018.
Download references
Acknowledgements
CC acknowledges the supervision offered by Professor Chris Hyde.
This publication forms a part of CC’s PhD. CC’s PhD was funded through the National Institute for Health Research (NIHR) Health Technology Assessment (HTA) Programme (Project Number 16/54/11). The open access fee for this publication was paid for by Exeter Medical School.
RG and NB were partially supported by the National Institute for Health Research (NIHR) Collaboration for Leadership in Applied Health Research and Care South West Peninsula.
The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health.
Author information
Authors and affiliations.
Institute of Health Research, University of Exeter Medical School, Exeter, UK
Chris Cooper & Jo Varley-Campbell
HEDS, School of Health and Related Research (ScHARR), University of Sheffield, Sheffield, UK
Andrew Booth
Nicky Britten
European Centre for Environment and Human Health, University of Exeter Medical School, Truro, UK
Ruth Garside
You can also search for this author in PubMed Google Scholar
Contributions
CC conceived the idea for this study and wrote the first draft of the manuscript. CC discussed this publication in PhD supervision with AB and separately with JVC. CC revised the publication with input and comments from AB, JVC, RG and NB. All authors revised the manuscript prior to submission. All authors read and approved the final manuscript.
Corresponding author
Correspondence to Chris Cooper .
Ethics declarations
Ethics approval and consent to participate, consent for publication, competing interests.
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Additional file
Additional file 1:.
Appendix tables and PubMed search strategy. Key studies used for pearl growing per key stage, working data extraction tables and the PubMed search strategy. (DOCX 30 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License ( http://creativecommons.org/licenses/by/4.0/ ), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated.
Reprints and permissions
About this article
Cite this article.
Cooper, C., Booth, A., Varley-Campbell, J. et al. Defining the process to literature searching in systematic reviews: a literature review of guidance and supporting studies. BMC Med Res Methodol 18 , 85 (2018). https://doi.org/10.1186/s12874-018-0545-3
Download citation
Received : 20 September 2017
Accepted : 06 August 2018
Published : 14 August 2018
DOI : https://doi.org/10.1186/s12874-018-0545-3
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Literature Search Process
- Citation Chasing
- Tacit Models
- Unique Guidance
- Information Specialists
BMC Medical Research Methodology
ISSN: 1471-2288
- General enquiries: [email protected]
An official website of the United States government
The .gov means it's official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you're on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- Browse Titles
NCBI Bookshelf. A service of the National Library of Medicine, National Institutes of Health.
Lau F, Kuziemsky C, editors. Handbook of eHealth Evaluation: An Evidence-based Approach [Internet]. Victoria (BC): University of Victoria; 2017 Feb 27.

Handbook of eHealth Evaluation: An Evidence-based Approach [Internet].
Chapter 9 methods for literature reviews.
Guy Paré and Spyros Kitsiou .
9.1. Introduction
Literature reviews play a critical role in scholarship because science remains, first and foremost, a cumulative endeavour ( vom Brocke et al., 2009 ). As in any academic discipline, rigorous knowledge syntheses are becoming indispensable in keeping up with an exponentially growing eHealth literature, assisting practitioners, academics, and graduate students in finding, evaluating, and synthesizing the contents of many empirical and conceptual papers. Among other methods, literature reviews are essential for: (a) identifying what has been written on a subject or topic; (b) determining the extent to which a specific research area reveals any interpretable trends or patterns; (c) aggregating empirical findings related to a narrow research question to support evidence-based practice; (d) generating new frameworks and theories; and (e) identifying topics or questions requiring more investigation ( Paré, Trudel, Jaana, & Kitsiou, 2015 ).
Literature reviews can take two major forms. The most prevalent one is the “literature review” or “background” section within a journal paper or a chapter in a graduate thesis. This section synthesizes the extant literature and usually identifies the gaps in knowledge that the empirical study addresses ( Sylvester, Tate, & Johnstone, 2013 ). It may also provide a theoretical foundation for the proposed study, substantiate the presence of the research problem, justify the research as one that contributes something new to the cumulated knowledge, or validate the methods and approaches for the proposed study ( Hart, 1998 ; Levy & Ellis, 2006 ).
The second form of literature review, which is the focus of this chapter, constitutes an original and valuable work of research in and of itself ( Paré et al., 2015 ). Rather than providing a base for a researcher’s own work, it creates a solid starting point for all members of the community interested in a particular area or topic ( Mulrow, 1987 ). The so-called “review article” is a journal-length paper which has an overarching purpose to synthesize the literature in a field, without collecting or analyzing any primary data ( Green, Johnson, & Adams, 2006 ).
When appropriately conducted, review articles represent powerful information sources for practitioners looking for state-of-the art evidence to guide their decision-making and work practices ( Paré et al., 2015 ). Further, high-quality reviews become frequently cited pieces of work which researchers seek out as a first clear outline of the literature when undertaking empirical studies ( Cooper, 1988 ; Rowe, 2014 ). Scholars who track and gauge the impact of articles have found that review papers are cited and downloaded more often than any other type of published article ( Cronin, Ryan, & Coughlan, 2008 ; Montori, Wilczynski, Morgan, Haynes, & Hedges, 2003 ; Patsopoulos, Analatos, & Ioannidis, 2005 ). The reason for their popularity may be the fact that reading the review enables one to have an overview, if not a detailed knowledge of the area in question, as well as references to the most useful primary sources ( Cronin et al., 2008 ). Although they are not easy to conduct, the commitment to complete a review article provides a tremendous service to one’s academic community ( Paré et al., 2015 ; Petticrew & Roberts, 2006 ). Most, if not all, peer-reviewed journals in the fields of medical informatics publish review articles of some type.
The main objectives of this chapter are fourfold: (a) to provide an overview of the major steps and activities involved in conducting a stand-alone literature review; (b) to describe and contrast the different types of review articles that can contribute to the eHealth knowledge base; (c) to illustrate each review type with one or two examples from the eHealth literature; and (d) to provide a series of recommendations for prospective authors of review articles in this domain.
9.2. Overview of the Literature Review Process and Steps
As explained in Templier and Paré (2015) , there are six generic steps involved in conducting a review article:
- formulating the research question(s) and objective(s),
- searching the extant literature,
- screening for inclusion,
- assessing the quality of primary studies,
- extracting data, and
- analyzing data.
Although these steps are presented here in sequential order, one must keep in mind that the review process can be iterative and that many activities can be initiated during the planning stage and later refined during subsequent phases ( Finfgeld-Connett & Johnson, 2013 ; Kitchenham & Charters, 2007 ).
Formulating the research question(s) and objective(s): As a first step, members of the review team must appropriately justify the need for the review itself ( Petticrew & Roberts, 2006 ), identify the review’s main objective(s) ( Okoli & Schabram, 2010 ), and define the concepts or variables at the heart of their synthesis ( Cooper & Hedges, 2009 ; Webster & Watson, 2002 ). Importantly, they also need to articulate the research question(s) they propose to investigate ( Kitchenham & Charters, 2007 ). In this regard, we concur with Jesson, Matheson, and Lacey (2011) that clearly articulated research questions are key ingredients that guide the entire review methodology; they underscore the type of information that is needed, inform the search for and selection of relevant literature, and guide or orient the subsequent analysis. Searching the extant literature: The next step consists of searching the literature and making decisions about the suitability of material to be considered in the review ( Cooper, 1988 ). There exist three main coverage strategies. First, exhaustive coverage means an effort is made to be as comprehensive as possible in order to ensure that all relevant studies, published and unpublished, are included in the review and, thus, conclusions are based on this all-inclusive knowledge base. The second type of coverage consists of presenting materials that are representative of most other works in a given field or area. Often authors who adopt this strategy will search for relevant articles in a small number of top-tier journals in a field ( Paré et al., 2015 ). In the third strategy, the review team concentrates on prior works that have been central or pivotal to a particular topic. This may include empirical studies or conceptual papers that initiated a line of investigation, changed how problems or questions were framed, introduced new methods or concepts, or engendered important debate ( Cooper, 1988 ). Screening for inclusion: The following step consists of evaluating the applicability of the material identified in the preceding step ( Levy & Ellis, 2006 ; vom Brocke et al., 2009 ). Once a group of potential studies has been identified, members of the review team must screen them to determine their relevance ( Petticrew & Roberts, 2006 ). A set of predetermined rules provides a basis for including or excluding certain studies. This exercise requires a significant investment on the part of researchers, who must ensure enhanced objectivity and avoid biases or mistakes. As discussed later in this chapter, for certain types of reviews there must be at least two independent reviewers involved in the screening process and a procedure to resolve disagreements must also be in place ( Liberati et al., 2009 ; Shea et al., 2009 ). Assessing the quality of primary studies: In addition to screening material for inclusion, members of the review team may need to assess the scientific quality of the selected studies, that is, appraise the rigour of the research design and methods. Such formal assessment, which is usually conducted independently by at least two coders, helps members of the review team refine which studies to include in the final sample, determine whether or not the differences in quality may affect their conclusions, or guide how they analyze the data and interpret the findings ( Petticrew & Roberts, 2006 ). Ascribing quality scores to each primary study or considering through domain-based evaluations which study components have or have not been designed and executed appropriately makes it possible to reflect on the extent to which the selected study addresses possible biases and maximizes validity ( Shea et al., 2009 ). Extracting data: The following step involves gathering or extracting applicable information from each primary study included in the sample and deciding what is relevant to the problem of interest ( Cooper & Hedges, 2009 ). Indeed, the type of data that should be recorded mainly depends on the initial research questions ( Okoli & Schabram, 2010 ). However, important information may also be gathered about how, when, where and by whom the primary study was conducted, the research design and methods, or qualitative/quantitative results ( Cooper & Hedges, 2009 ). Analyzing and synthesizing data : As a final step, members of the review team must collate, summarize, aggregate, organize, and compare the evidence extracted from the included studies. The extracted data must be presented in a meaningful way that suggests a new contribution to the extant literature ( Jesson et al., 2011 ). Webster and Watson (2002) warn researchers that literature reviews should be much more than lists of papers and should provide a coherent lens to make sense of extant knowledge on a given topic. There exist several methods and techniques for synthesizing quantitative (e.g., frequency analysis, meta-analysis) and qualitative (e.g., grounded theory, narrative analysis, meta-ethnography) evidence ( Dixon-Woods, Agarwal, Jones, Young, & Sutton, 2005 ; Thomas & Harden, 2008 ).
9.3. Types of Review Articles and Brief Illustrations
EHealth researchers have at their disposal a number of approaches and methods for making sense out of existing literature, all with the purpose of casting current research findings into historical contexts or explaining contradictions that might exist among a set of primary research studies conducted on a particular topic. Our classification scheme is largely inspired from Paré and colleagues’ (2015) typology. Below we present and illustrate those review types that we feel are central to the growth and development of the eHealth domain.
9.3.1. Narrative Reviews
The narrative review is the “traditional” way of reviewing the extant literature and is skewed towards a qualitative interpretation of prior knowledge ( Sylvester et al., 2013 ). Put simply, a narrative review attempts to summarize or synthesize what has been written on a particular topic but does not seek generalization or cumulative knowledge from what is reviewed ( Davies, 2000 ; Green et al., 2006 ). Instead, the review team often undertakes the task of accumulating and synthesizing the literature to demonstrate the value of a particular point of view ( Baumeister & Leary, 1997 ). As such, reviewers may selectively ignore or limit the attention paid to certain studies in order to make a point. In this rather unsystematic approach, the selection of information from primary articles is subjective, lacks explicit criteria for inclusion and can lead to biased interpretations or inferences ( Green et al., 2006 ). There are several narrative reviews in the particular eHealth domain, as in all fields, which follow such an unstructured approach ( Silva et al., 2015 ; Paul et al., 2015 ).
Despite these criticisms, this type of review can be very useful in gathering together a volume of literature in a specific subject area and synthesizing it. As mentioned above, its primary purpose is to provide the reader with a comprehensive background for understanding current knowledge and highlighting the significance of new research ( Cronin et al., 2008 ). Faculty like to use narrative reviews in the classroom because they are often more up to date than textbooks, provide a single source for students to reference, and expose students to peer-reviewed literature ( Green et al., 2006 ). For researchers, narrative reviews can inspire research ideas by identifying gaps or inconsistencies in a body of knowledge, thus helping researchers to determine research questions or formulate hypotheses. Importantly, narrative reviews can also be used as educational articles to bring practitioners up to date with certain topics of issues ( Green et al., 2006 ).
Recently, there have been several efforts to introduce more rigour in narrative reviews that will elucidate common pitfalls and bring changes into their publication standards. Information systems researchers, among others, have contributed to advancing knowledge on how to structure a “traditional” review. For instance, Levy and Ellis (2006) proposed a generic framework for conducting such reviews. Their model follows the systematic data processing approach comprised of three steps, namely: (a) literature search and screening; (b) data extraction and analysis; and (c) writing the literature review. They provide detailed and very helpful instructions on how to conduct each step of the review process. As another methodological contribution, vom Brocke et al. (2009) offered a series of guidelines for conducting literature reviews, with a particular focus on how to search and extract the relevant body of knowledge. Last, Bandara, Miskon, and Fielt (2011) proposed a structured, predefined and tool-supported method to identify primary studies within a feasible scope, extract relevant content from identified articles, synthesize and analyze the findings, and effectively write and present the results of the literature review. We highly recommend that prospective authors of narrative reviews consult these useful sources before embarking on their work.
Darlow and Wen (2015) provide a good example of a highly structured narrative review in the eHealth field. These authors synthesized published articles that describe the development process of mobile health ( m-health ) interventions for patients’ cancer care self-management. As in most narrative reviews, the scope of the research questions being investigated is broad: (a) how development of these systems are carried out; (b) which methods are used to investigate these systems; and (c) what conclusions can be drawn as a result of the development of these systems. To provide clear answers to these questions, a literature search was conducted on six electronic databases and Google Scholar . The search was performed using several terms and free text words, combining them in an appropriate manner. Four inclusion and three exclusion criteria were utilized during the screening process. Both authors independently reviewed each of the identified articles to determine eligibility and extract study information. A flow diagram shows the number of studies identified, screened, and included or excluded at each stage of study selection. In terms of contributions, this review provides a series of practical recommendations for m-health intervention development.
9.3.2. Descriptive or Mapping Reviews
The primary goal of a descriptive review is to determine the extent to which a body of knowledge in a particular research topic reveals any interpretable pattern or trend with respect to pre-existing propositions, theories, methodologies or findings ( King & He, 2005 ; Paré et al., 2015 ). In contrast with narrative reviews, descriptive reviews follow a systematic and transparent procedure, including searching, screening and classifying studies ( Petersen, Vakkalanka, & Kuzniarz, 2015 ). Indeed, structured search methods are used to form a representative sample of a larger group of published works ( Paré et al., 2015 ). Further, authors of descriptive reviews extract from each study certain characteristics of interest, such as publication year, research methods, data collection techniques, and direction or strength of research outcomes (e.g., positive, negative, or non-significant) in the form of frequency analysis to produce quantitative results ( Sylvester et al., 2013 ). In essence, each study included in a descriptive review is treated as the unit of analysis and the published literature as a whole provides a database from which the authors attempt to identify any interpretable trends or draw overall conclusions about the merits of existing conceptualizations, propositions, methods or findings ( Paré et al., 2015 ). In doing so, a descriptive review may claim that its findings represent the state of the art in a particular domain ( King & He, 2005 ).
In the fields of health sciences and medical informatics, reviews that focus on examining the range, nature and evolution of a topic area are described by Anderson, Allen, Peckham, and Goodwin (2008) as mapping reviews . Like descriptive reviews, the research questions are generic and usually relate to publication patterns and trends. There is no preconceived plan to systematically review all of the literature although this can be done. Instead, researchers often present studies that are representative of most works published in a particular area and they consider a specific time frame to be mapped.
An example of this approach in the eHealth domain is offered by DeShazo, Lavallie, and Wolf (2009). The purpose of this descriptive or mapping review was to characterize publication trends in the medical informatics literature over a 20-year period (1987 to 2006). To achieve this ambitious objective, the authors performed a bibliometric analysis of medical informatics citations indexed in medline using publication trends, journal frequencies, impact factors, Medical Subject Headings (MeSH) term frequencies, and characteristics of citations. Findings revealed that there were over 77,000 medical informatics articles published during the covered period in numerous journals and that the average annual growth rate was 12%. The MeSH term analysis also suggested a strong interdisciplinary trend. Finally, average impact scores increased over time with two notable growth periods. Overall, patterns in research outputs that seem to characterize the historic trends and current components of the field of medical informatics suggest it may be a maturing discipline (DeShazo et al., 2009).
9.3.3. Scoping Reviews
Scoping reviews attempt to provide an initial indication of the potential size and nature of the extant literature on an emergent topic (Arksey & O’Malley, 2005; Daudt, van Mossel, & Scott, 2013 ; Levac, Colquhoun, & O’Brien, 2010). A scoping review may be conducted to examine the extent, range and nature of research activities in a particular area, determine the value of undertaking a full systematic review (discussed next), or identify research gaps in the extant literature ( Paré et al., 2015 ). In line with their main objective, scoping reviews usually conclude with the presentation of a detailed research agenda for future works along with potential implications for both practice and research.
Unlike narrative and descriptive reviews, the whole point of scoping the field is to be as comprehensive as possible, including grey literature (Arksey & O’Malley, 2005). Inclusion and exclusion criteria must be established to help researchers eliminate studies that are not aligned with the research questions. It is also recommended that at least two independent coders review abstracts yielded from the search strategy and then the full articles for study selection ( Daudt et al., 2013 ). The synthesized evidence from content or thematic analysis is relatively easy to present in tabular form (Arksey & O’Malley, 2005; Thomas & Harden, 2008 ).
One of the most highly cited scoping reviews in the eHealth domain was published by Archer, Fevrier-Thomas, Lokker, McKibbon, and Straus (2011) . These authors reviewed the existing literature on personal health record ( phr ) systems including design, functionality, implementation, applications, outcomes, and benefits. Seven databases were searched from 1985 to March 2010. Several search terms relating to phr s were used during this process. Two authors independently screened titles and abstracts to determine inclusion status. A second screen of full-text articles, again by two independent members of the research team, ensured that the studies described phr s. All in all, 130 articles met the criteria and their data were extracted manually into a database. The authors concluded that although there is a large amount of survey, observational, cohort/panel, and anecdotal evidence of phr benefits and satisfaction for patients, more research is needed to evaluate the results of phr implementations. Their in-depth analysis of the literature signalled that there is little solid evidence from randomized controlled trials or other studies through the use of phr s. Hence, they suggested that more research is needed that addresses the current lack of understanding of optimal functionality and usability of these systems, and how they can play a beneficial role in supporting patient self-management ( Archer et al., 2011 ).

9.3.4. Forms of Aggregative Reviews
Healthcare providers, practitioners, and policy-makers are nowadays overwhelmed with large volumes of information, including research-based evidence from numerous clinical trials and evaluation studies, assessing the effectiveness of health information technologies and interventions ( Ammenwerth & de Keizer, 2004 ; Deshazo et al., 2009 ). It is unrealistic to expect that all these disparate actors will have the time, skills, and necessary resources to identify the available evidence in the area of their expertise and consider it when making decisions. Systematic reviews that involve the rigorous application of scientific strategies aimed at limiting subjectivity and bias (i.e., systematic and random errors) can respond to this challenge.
Systematic reviews attempt to aggregate, appraise, and synthesize in a single source all empirical evidence that meet a set of previously specified eligibility criteria in order to answer a clearly formulated and often narrow research question on a particular topic of interest to support evidence-based practice ( Liberati et al., 2009 ). They adhere closely to explicit scientific principles ( Liberati et al., 2009 ) and rigorous methodological guidelines (Higgins & Green, 2008) aimed at reducing random and systematic errors that can lead to deviations from the truth in results or inferences. The use of explicit methods allows systematic reviews to aggregate a large body of research evidence, assess whether effects or relationships are in the same direction and of the same general magnitude, explain possible inconsistencies between study results, and determine the strength of the overall evidence for every outcome of interest based on the quality of included studies and the general consistency among them ( Cook, Mulrow, & Haynes, 1997 ). The main procedures of a systematic review involve:
- Formulating a review question and developing a search strategy based on explicit inclusion criteria for the identification of eligible studies (usually described in the context of a detailed review protocol).
- Searching for eligible studies using multiple databases and information sources, including grey literature sources, without any language restrictions.
- Selecting studies, extracting data, and assessing risk of bias in a duplicate manner using two independent reviewers to avoid random or systematic errors in the process.
- Analyzing data using quantitative or qualitative methods.
- Presenting results in summary of findings tables.
- Interpreting results and drawing conclusions.
Many systematic reviews, but not all, use statistical methods to combine the results of independent studies into a single quantitative estimate or summary effect size. Known as meta-analyses , these reviews use specific data extraction and statistical techniques (e.g., network, frequentist, or Bayesian meta-analyses) to calculate from each study by outcome of interest an effect size along with a confidence interval that reflects the degree of uncertainty behind the point estimate of effect ( Borenstein, Hedges, Higgins, & Rothstein, 2009 ; Deeks, Higgins, & Altman, 2008 ). Subsequently, they use fixed or random-effects analysis models to combine the results of the included studies, assess statistical heterogeneity, and calculate a weighted average of the effect estimates from the different studies, taking into account their sample sizes. The summary effect size is a value that reflects the average magnitude of the intervention effect for a particular outcome of interest or, more generally, the strength of a relationship between two variables across all studies included in the systematic review. By statistically combining data from multiple studies, meta-analyses can create more precise and reliable estimates of intervention effects than those derived from individual studies alone, when these are examined independently as discrete sources of information.
The review by Gurol-Urganci, de Jongh, Vodopivec-Jamsek, Atun, and Car (2013) on the effects of mobile phone messaging reminders for attendance at healthcare appointments is an illustrative example of a high-quality systematic review with meta-analysis. Missed appointments are a major cause of inefficiency in healthcare delivery with substantial monetary costs to health systems. These authors sought to assess whether mobile phone-based appointment reminders delivered through Short Message Service ( sms ) or Multimedia Messaging Service ( mms ) are effective in improving rates of patient attendance and reducing overall costs. To this end, they conducted a comprehensive search on multiple databases using highly sensitive search strategies without language or publication-type restrictions to identify all rct s that are eligible for inclusion. In order to minimize the risk of omitting eligible studies not captured by the original search, they supplemented all electronic searches with manual screening of trial registers and references contained in the included studies. Study selection, data extraction, and risk of bias assessments were performed independently by two coders using standardized methods to ensure consistency and to eliminate potential errors. Findings from eight rct s involving 6,615 participants were pooled into meta-analyses to calculate the magnitude of effects that mobile text message reminders have on the rate of attendance at healthcare appointments compared to no reminders and phone call reminders.
Meta-analyses are regarded as powerful tools for deriving meaningful conclusions. However, there are situations in which it is neither reasonable nor appropriate to pool studies together using meta-analytic methods simply because there is extensive clinical heterogeneity between the included studies or variation in measurement tools, comparisons, or outcomes of interest. In these cases, systematic reviews can use qualitative synthesis methods such as vote counting, content analysis, classification schemes and tabulations, as an alternative approach to narratively synthesize the results of the independent studies included in the review. This form of review is known as qualitative systematic review.
A rigorous example of one such review in the eHealth domain is presented by Mickan, Atherton, Roberts, Heneghan, and Tilson (2014) on the use of handheld computers by healthcare professionals and their impact on access to information and clinical decision-making. In line with the methodological guidelines for systematic reviews, these authors: (a) developed and registered with prospero ( www.crd.york.ac.uk/ prospero / ) an a priori review protocol; (b) conducted comprehensive searches for eligible studies using multiple databases and other supplementary strategies (e.g., forward searches); and (c) subsequently carried out study selection, data extraction, and risk of bias assessments in a duplicate manner to eliminate potential errors in the review process. Heterogeneity between the included studies in terms of reported outcomes and measures precluded the use of meta-analytic methods. To this end, the authors resorted to using narrative analysis and synthesis to describe the effectiveness of handheld computers on accessing information for clinical knowledge, adherence to safety and clinical quality guidelines, and diagnostic decision-making.
In recent years, the number of systematic reviews in the field of health informatics has increased considerably. Systematic reviews with discordant findings can cause great confusion and make it difficult for decision-makers to interpret the review-level evidence ( Moher, 2013 ). Therefore, there is a growing need for appraisal and synthesis of prior systematic reviews to ensure that decision-making is constantly informed by the best available accumulated evidence. Umbrella reviews , also known as overviews of systematic reviews, are tertiary types of evidence synthesis that aim to accomplish this; that is, they aim to compare and contrast findings from multiple systematic reviews and meta-analyses ( Becker & Oxman, 2008 ). Umbrella reviews generally adhere to the same principles and rigorous methodological guidelines used in systematic reviews. However, the unit of analysis in umbrella reviews is the systematic review rather than the primary study ( Becker & Oxman, 2008 ). Unlike systematic reviews that have a narrow focus of inquiry, umbrella reviews focus on broader research topics for which there are several potential interventions ( Smith, Devane, Begley, & Clarke, 2011 ). A recent umbrella review on the effects of home telemonitoring interventions for patients with heart failure critically appraised, compared, and synthesized evidence from 15 systematic reviews to investigate which types of home telemonitoring technologies and forms of interventions are more effective in reducing mortality and hospital admissions ( Kitsiou, Paré, & Jaana, 2015 ).
9.3.5. Realist Reviews
Realist reviews are theory-driven interpretative reviews developed to inform, enhance, or supplement conventional systematic reviews by making sense of heterogeneous evidence about complex interventions applied in diverse contexts in a way that informs policy decision-making ( Greenhalgh, Wong, Westhorp, & Pawson, 2011 ). They originated from criticisms of positivist systematic reviews which centre on their “simplistic” underlying assumptions ( Oates, 2011 ). As explained above, systematic reviews seek to identify causation. Such logic is appropriate for fields like medicine and education where findings of randomized controlled trials can be aggregated to see whether a new treatment or intervention does improve outcomes. However, many argue that it is not possible to establish such direct causal links between interventions and outcomes in fields such as social policy, management, and information systems where for any intervention there is unlikely to be a regular or consistent outcome ( Oates, 2011 ; Pawson, 2006 ; Rousseau, Manning, & Denyer, 2008 ).
To circumvent these limitations, Pawson, Greenhalgh, Harvey, and Walshe (2005) have proposed a new approach for synthesizing knowledge that seeks to unpack the mechanism of how “complex interventions” work in particular contexts. The basic research question — what works? — which is usually associated with systematic reviews changes to: what is it about this intervention that works, for whom, in what circumstances, in what respects and why? Realist reviews have no particular preference for either quantitative or qualitative evidence. As a theory-building approach, a realist review usually starts by articulating likely underlying mechanisms and then scrutinizes available evidence to find out whether and where these mechanisms are applicable ( Shepperd et al., 2009 ). Primary studies found in the extant literature are viewed as case studies which can test and modify the initial theories ( Rousseau et al., 2008 ).
The main objective pursued in the realist review conducted by Otte-Trojel, de Bont, Rundall, and van de Klundert (2014) was to examine how patient portals contribute to health service delivery and patient outcomes. The specific goals were to investigate how outcomes are produced and, most importantly, how variations in outcomes can be explained. The research team started with an exploratory review of background documents and research studies to identify ways in which patient portals may contribute to health service delivery and patient outcomes. The authors identified six main ways which represent “educated guesses” to be tested against the data in the evaluation studies. These studies were identified through a formal and systematic search in four databases between 2003 and 2013. Two members of the research team selected the articles using a pre-established list of inclusion and exclusion criteria and following a two-step procedure. The authors then extracted data from the selected articles and created several tables, one for each outcome category. They organized information to bring forward those mechanisms where patient portals contribute to outcomes and the variation in outcomes across different contexts.
9.3.6. Critical Reviews
Lastly, critical reviews aim to provide a critical evaluation and interpretive analysis of existing literature on a particular topic of interest to reveal strengths, weaknesses, contradictions, controversies, inconsistencies, and/or other important issues with respect to theories, hypotheses, research methods or results ( Baumeister & Leary, 1997 ; Kirkevold, 1997 ). Unlike other review types, critical reviews attempt to take a reflective account of the research that has been done in a particular area of interest, and assess its credibility by using appraisal instruments or critical interpretive methods. In this way, critical reviews attempt to constructively inform other scholars about the weaknesses of prior research and strengthen knowledge development by giving focus and direction to studies for further improvement ( Kirkevold, 1997 ).
Kitsiou, Paré, and Jaana (2013) provide an example of a critical review that assessed the methodological quality of prior systematic reviews of home telemonitoring studies for chronic patients. The authors conducted a comprehensive search on multiple databases to identify eligible reviews and subsequently used a validated instrument to conduct an in-depth quality appraisal. Results indicate that the majority of systematic reviews in this particular area suffer from important methodological flaws and biases that impair their internal validity and limit their usefulness for clinical and decision-making purposes. To this end, they provide a number of recommendations to strengthen knowledge development towards improving the design and execution of future reviews on home telemonitoring.
9.4. Summary
Table 9.1 outlines the main types of literature reviews that were described in the previous sub-sections and summarizes the main characteristics that distinguish one review type from another. It also includes key references to methodological guidelines and useful sources that can be used by eHealth scholars and researchers for planning and developing reviews.
Typology of Literature Reviews (adapted from Paré et al., 2015).
As shown in Table 9.1 , each review type addresses different kinds of research questions or objectives, which subsequently define and dictate the methods and approaches that need to be used to achieve the overarching goal(s) of the review. For example, in the case of narrative reviews, there is greater flexibility in searching and synthesizing articles ( Green et al., 2006 ). Researchers are often relatively free to use a diversity of approaches to search, identify, and select relevant scientific articles, describe their operational characteristics, present how the individual studies fit together, and formulate conclusions. On the other hand, systematic reviews are characterized by their high level of systematicity, rigour, and use of explicit methods, based on an “a priori” review plan that aims to minimize bias in the analysis and synthesis process (Higgins & Green, 2008). Some reviews are exploratory in nature (e.g., scoping/mapping reviews), whereas others may be conducted to discover patterns (e.g., descriptive reviews) or involve a synthesis approach that may include the critical analysis of prior research ( Paré et al., 2015 ). Hence, in order to select the most appropriate type of review, it is critical to know before embarking on a review project, why the research synthesis is conducted and what type of methods are best aligned with the pursued goals.
9.5. Concluding Remarks
In light of the increased use of evidence-based practice and research generating stronger evidence ( Grady et al., 2011 ; Lyden et al., 2013 ), review articles have become essential tools for summarizing, synthesizing, integrating or critically appraising prior knowledge in the eHealth field. As mentioned earlier, when rigorously conducted review articles represent powerful information sources for eHealth scholars and practitioners looking for state-of-the-art evidence. The typology of literature reviews we used herein will allow eHealth researchers, graduate students and practitioners to gain a better understanding of the similarities and differences between review types.
We must stress that this classification scheme does not privilege any specific type of review as being of higher quality than another ( Paré et al., 2015 ). As explained above, each type of review has its own strengths and limitations. Having said that, we realize that the methodological rigour of any review — be it qualitative, quantitative or mixed — is a critical aspect that should be considered seriously by prospective authors. In the present context, the notion of rigour refers to the reliability and validity of the review process described in section 9.2. For one thing, reliability is related to the reproducibility of the review process and steps, which is facilitated by a comprehensive documentation of the literature search process, extraction, coding and analysis performed in the review. Whether the search is comprehensive or not, whether it involves a methodical approach for data extraction and synthesis or not, it is important that the review documents in an explicit and transparent manner the steps and approach that were used in the process of its development. Next, validity characterizes the degree to which the review process was conducted appropriately. It goes beyond documentation and reflects decisions related to the selection of the sources, the search terms used, the period of time covered, the articles selected in the search, and the application of backward and forward searches ( vom Brocke et al., 2009 ). In short, the rigour of any review article is reflected by the explicitness of its methods (i.e., transparency) and the soundness of the approach used. We refer those interested in the concepts of rigour and quality to the work of Templier and Paré (2015) which offers a detailed set of methodological guidelines for conducting and evaluating various types of review articles.
To conclude, our main objective in this chapter was to demystify the various types of literature reviews that are central to the continuous development of the eHealth field. It is our hope that our descriptive account will serve as a valuable source for those conducting, evaluating or using reviews in this important and growing domain.
- Ammenwerth E., de Keizer N. An inventory of evaluation studies of information technology in health care. Trends in evaluation research, 1982-2002. International Journal of Medical Informatics. 2004; 44 (1):44–56. [ PubMed : 15778794 ]
- Anderson S., Allen P., Peckham S., Goodwin N. Asking the right questions: scoping studies in the commissioning of research on the organisation and delivery of health services. Health Research Policy and Systems. 2008; 6 (7):1–12. [ PMC free article : PMC2500008 ] [ PubMed : 18613961 ] [ CrossRef ]
- Archer N., Fevrier-Thomas U., Lokker C., McKibbon K. A., Straus S.E. Personal health records: a scoping review. Journal of American Medical Informatics Association. 2011; 18 (4):515–522. [ PMC free article : PMC3128401 ] [ PubMed : 21672914 ]
- Arksey H., O’Malley L. Scoping studies: towards a methodological framework. International Journal of Social Research Methodology. 2005; 8 (1):19–32.
- A systematic, tool-supported method for conducting literature reviews in information systems. Paper presented at the Proceedings of the 19th European Conference on Information Systems ( ecis 2011); June 9 to 11; Helsinki, Finland. 2011.
- Baumeister R. F., Leary M.R. Writing narrative literature reviews. Review of General Psychology. 1997; 1 (3):311–320.
- Becker L. A., Oxman A.D. In: Cochrane handbook for systematic reviews of interventions. Higgins J. P. T., Green S., editors. Hoboken, nj : John Wiley & Sons, Ltd; 2008. Overviews of reviews; pp. 607–631.
- Borenstein M., Hedges L., Higgins J., Rothstein H. Introduction to meta-analysis. Hoboken, nj : John Wiley & Sons Inc; 2009.
- Cook D. J., Mulrow C. D., Haynes B. Systematic reviews: Synthesis of best evidence for clinical decisions. Annals of Internal Medicine. 1997; 126 (5):376–380. [ PubMed : 9054282 ]
- Cooper H., Hedges L.V. In: The handbook of research synthesis and meta-analysis. 2nd ed. Cooper H., Hedges L. V., Valentine J. C., editors. New York: Russell Sage Foundation; 2009. Research synthesis as a scientific process; pp. 3–17.
- Cooper H. M. Organizing knowledge syntheses: A taxonomy of literature reviews. Knowledge in Society. 1988; 1 (1):104–126.
- Cronin P., Ryan F., Coughlan M. Undertaking a literature review: a step-by-step approach. British Journal of Nursing. 2008; 17 (1):38–43. [ PubMed : 18399395 ]
- Darlow S., Wen K.Y. Development testing of mobile health interventions for cancer patient self-management: A review. Health Informatics Journal. 2015 (online before print). [ PubMed : 25916831 ] [ CrossRef ]
- Daudt H. M., van Mossel C., Scott S.J. Enhancing the scoping study methodology: a large, inter-professional team’s experience with Arksey and O’Malley’s framework. bmc Medical Research Methodology. 2013; 13 :48. [ PMC free article : PMC3614526 ] [ PubMed : 23522333 ] [ CrossRef ]
- Davies P. The relevance of systematic reviews to educational policy and practice. Oxford Review of Education. 2000; 26 (3-4):365–378.
- Deeks J. J., Higgins J. P. T., Altman D.G. In: Cochrane handbook for systematic reviews of interventions. Higgins J. P. T., Green S., editors. Hoboken, nj : John Wiley & Sons, Ltd; 2008. Analysing data and undertaking meta-analyses; pp. 243–296.
- Deshazo J. P., Lavallie D. L., Wolf F.M. Publication trends in the medical informatics literature: 20 years of “Medical Informatics” in mesh . bmc Medical Informatics and Decision Making. 2009; 9 :7. [ PMC free article : PMC2652453 ] [ PubMed : 19159472 ] [ CrossRef ]
- Dixon-Woods M., Agarwal S., Jones D., Young B., Sutton A. Synthesising qualitative and quantitative evidence: a review of possible methods. Journal of Health Services Research and Policy. 2005; 10 (1):45–53. [ PubMed : 15667704 ]
- Finfgeld-Connett D., Johnson E.D. Literature search strategies for conducting knowledge-building and theory-generating qualitative systematic reviews. Journal of Advanced Nursing. 2013; 69 (1):194–204. [ PMC free article : PMC3424349 ] [ PubMed : 22591030 ]
- Grady B., Myers K. M., Nelson E. L., Belz N., Bennett L., Carnahan L. … Guidelines Working Group. Evidence-based practice for telemental health. Telemedicine Journal and E Health. 2011; 17 (2):131–148. [ PubMed : 21385026 ]
- Green B. N., Johnson C. D., Adams A. Writing narrative literature reviews for peer-reviewed journals: secrets of the trade. Journal of Chiropractic Medicine. 2006; 5 (3):101–117. [ PMC free article : PMC2647067 ] [ PubMed : 19674681 ]
- Greenhalgh T., Wong G., Westhorp G., Pawson R. Protocol–realist and meta-narrative evidence synthesis: evolving standards ( rameses ). bmc Medical Research Methodology. 2011; 11 :115. [ PMC free article : PMC3173389 ] [ PubMed : 21843376 ]
- Gurol-Urganci I., de Jongh T., Vodopivec-Jamsek V., Atun R., Car J. Mobile phone messaging reminders for attendance at healthcare appointments. Cochrane Database System Review. 2013; 12 cd 007458. [ PMC free article : PMC6485985 ] [ PubMed : 24310741 ] [ CrossRef ]
- Hart C. Doing a literature review: Releasing the social science research imagination. London: SAGE Publications; 1998.
- Higgins J. P. T., Green S., editors. Cochrane handbook for systematic reviews of interventions: Cochrane book series. Hoboken, nj : Wiley-Blackwell; 2008.
- Jesson J., Matheson L., Lacey F.M. Doing your literature review: traditional and systematic techniques. Los Angeles & London: SAGE Publications; 2011.
- King W. R., He J. Understanding the role and methods of meta-analysis in IS research. Communications of the Association for Information Systems. 2005; 16 :1.
- Kirkevold M. Integrative nursing research — an important strategy to further the development of nursing science and nursing practice. Journal of Advanced Nursing. 1997; 25 (5):977–984. [ PubMed : 9147203 ]
- Kitchenham B., Charters S. ebse Technical Report Version 2.3. Keele & Durham. uk : Keele University & University of Durham; 2007. Guidelines for performing systematic literature reviews in software engineering.
- Kitsiou S., Paré G., Jaana M. Systematic reviews and meta-analyses of home telemonitoring interventions for patients with chronic diseases: a critical assessment of their methodological quality. Journal of Medical Internet Research. 2013; 15 (7):e150. [ PMC free article : PMC3785977 ] [ PubMed : 23880072 ]
- Kitsiou S., Paré G., Jaana M. Effects of home telemonitoring interventions on patients with chronic heart failure: an overview of systematic reviews. Journal of Medical Internet Research. 2015; 17 (3):e63. [ PMC free article : PMC4376138 ] [ PubMed : 25768664 ]
- Levac D., Colquhoun H., O’Brien K. K. Scoping studies: advancing the methodology. Implementation Science. 2010; 5 (1):69. [ PMC free article : PMC2954944 ] [ PubMed : 20854677 ]
- Levy Y., Ellis T.J. A systems approach to conduct an effective literature review in support of information systems research. Informing Science. 2006; 9 :181–211.
- Liberati A., Altman D. G., Tetzlaff J., Mulrow C., Gøtzsche P. C., Ioannidis J. P. A. et al. Moher D. The prisma statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. Annals of Internal Medicine. 2009; 151 (4):W-65. [ PubMed : 19622512 ]
- Lyden J. R., Zickmund S. L., Bhargava T. D., Bryce C. L., Conroy M. B., Fischer G. S. et al. McTigue K. M. Implementing health information technology in a patient-centered manner: Patient experiences with an online evidence-based lifestyle intervention. Journal for Healthcare Quality. 2013; 35 (5):47–57. [ PubMed : 24004039 ]
- Mickan S., Atherton H., Roberts N. W., Heneghan C., Tilson J.K. Use of handheld computers in clinical practice: a systematic review. bmc Medical Informatics and Decision Making. 2014; 14 :56. [ PMC free article : PMC4099138 ] [ PubMed : 24998515 ]
- Moher D. The problem of duplicate systematic reviews. British Medical Journal. 2013; 347 (5040) [ PubMed : 23945367 ] [ CrossRef ]
- Montori V. M., Wilczynski N. L., Morgan D., Haynes R. B., Hedges T. Systematic reviews: a cross-sectional study of location and citation counts. bmc Medicine. 2003; 1 :2. [ PMC free article : PMC281591 ] [ PubMed : 14633274 ]
- Mulrow C. D. The medical review article: state of the science. Annals of Internal Medicine. 1987; 106 (3):485–488. [ PubMed : 3813259 ] [ CrossRef ]
- Evidence-based information systems: A decade later. Proceedings of the European Conference on Information Systems ; 2011. Retrieved from http://aisel .aisnet.org/cgi/viewcontent .cgi?article =1221&context =ecis2011 .
- Okoli C., Schabram K. A guide to conducting a systematic literature review of information systems research. ssrn Electronic Journal. 2010
- Otte-Trojel T., de Bont A., Rundall T. G., van de Klundert J. How outcomes are achieved through patient portals: a realist review. Journal of American Medical Informatics Association. 2014; 21 (4):751–757. [ PMC free article : PMC4078283 ] [ PubMed : 24503882 ]
- Paré G., Trudel M.-C., Jaana M., Kitsiou S. Synthesizing information systems knowledge: A typology of literature reviews. Information & Management. 2015; 52 (2):183–199.
- Patsopoulos N. A., Analatos A. A., Ioannidis J.P. A. Relative citation impact of various study designs in the health sciences. Journal of the American Medical Association. 2005; 293 (19):2362–2366. [ PubMed : 15900006 ]
- Paul M. M., Greene C. M., Newton-Dame R., Thorpe L. E., Perlman S. E., McVeigh K. H., Gourevitch M.N. The state of population health surveillance using electronic health records: A narrative review. Population Health Management. 2015; 18 (3):209–216. [ PubMed : 25608033 ]
- Pawson R. Evidence-based policy: a realist perspective. London: SAGE Publications; 2006.
- Pawson R., Greenhalgh T., Harvey G., Walshe K. Realist review—a new method of systematic review designed for complex policy interventions. Journal of Health Services Research & Policy. 2005; 10 (Suppl 1):21–34. [ PubMed : 16053581 ]
- Petersen K., Vakkalanka S., Kuzniarz L. Guidelines for conducting systematic mapping studies in software engineering: An update. Information and Software Technology. 2015; 64 :1–18.
- Petticrew M., Roberts H. Systematic reviews in the social sciences: A practical guide. Malden, ma : Blackwell Publishing Co; 2006.
- Rousseau D. M., Manning J., Denyer D. Evidence in management and organizational science: Assembling the field’s full weight of scientific knowledge through syntheses. The Academy of Management Annals. 2008; 2 (1):475–515.
- Rowe F. What literature review is not: diversity, boundaries and recommendations. European Journal of Information Systems. 2014; 23 (3):241–255.
- Shea B. J., Hamel C., Wells G. A., Bouter L. M., Kristjansson E., Grimshaw J. et al. Boers M. amstar is a reliable and valid measurement tool to assess the methodological quality of systematic reviews. Journal of Clinical Epidemiology. 2009; 62 (10):1013–1020. [ PubMed : 19230606 ]
- Shepperd S., Lewin S., Straus S., Clarke M., Eccles M. P., Fitzpatrick R. et al. Sheikh A. Can we systematically review studies that evaluate complex interventions? PLoS Medicine. 2009; 6 (8):e1000086. [ PMC free article : PMC2717209 ] [ PubMed : 19668360 ]
- Silva B. M., Rodrigues J. J., de la Torre Díez I., López-Coronado M., Saleem K. Mobile-health: A review of current state in 2015. Journal of Biomedical Informatics. 2015; 56 :265–272. [ PubMed : 26071682 ]
- Smith V., Devane D., Begley C., Clarke M. Methodology in conducting a systematic review of systematic reviews of healthcare interventions. bmc Medical Research Methodology. 2011; 11 (1):15. [ PMC free article : PMC3039637 ] [ PubMed : 21291558 ]
- Sylvester A., Tate M., Johnstone D. Beyond synthesis: re-presenting heterogeneous research literature. Behaviour & Information Technology. 2013; 32 (12):1199–1215.
- Templier M., Paré G. A framework for guiding and evaluating literature reviews. Communications of the Association for Information Systems. 2015; 37 (6):112–137.
- Thomas J., Harden A. Methods for the thematic synthesis of qualitative research in systematic reviews. bmc Medical Research Methodology. 2008; 8 (1):45. [ PMC free article : PMC2478656 ] [ PubMed : 18616818 ]
- Reconstructing the giant: on the importance of rigour in documenting the literature search process. Paper presented at the Proceedings of the 17th European Conference on Information Systems ( ecis 2009); Verona, Italy. 2009.
- Webster J., Watson R.T. Analyzing the past to prepare for the future: Writing a literature review. Management Information Systems Quarterly. 2002; 26 (2):11.
- Whitlock E. P., Lin J. S., Chou R., Shekelle P., Robinson K.A. Using existing systematic reviews in complex systematic reviews. Annals of Internal Medicine. 2008; 148 (10):776–782. [ PubMed : 18490690 ]
This publication is licensed under a Creative Commons License, Attribution-Noncommercial 4.0 International License (CC BY-NC 4.0): see https://creativecommons.org/licenses/by-nc/4.0/
- Cite this Page Paré G, Kitsiou S. Chapter 9 Methods for Literature Reviews. In: Lau F, Kuziemsky C, editors. Handbook of eHealth Evaluation: An Evidence-based Approach [Internet]. Victoria (BC): University of Victoria; 2017 Feb 27.
- PDF version of this title (4.5M)
- Disable Glossary Links
In this Page
- Introduction
- Overview of the Literature Review Process and Steps
- Types of Review Articles and Brief Illustrations
- Concluding Remarks
Related information
- PMC PubMed Central citations
- PubMed Links to PubMed
Recent Activity
- Chapter 9 Methods for Literature Reviews - Handbook of eHealth Evaluation: An Ev... Chapter 9 Methods for Literature Reviews - Handbook of eHealth Evaluation: An Evidence-based Approach
Your browsing activity is empty.
Activity recording is turned off.
Turn recording back on
Connect with NLM
National Library of Medicine 8600 Rockville Pike Bethesda, MD 20894
Web Policies FOIA HHS Vulnerability Disclosure
Help Accessibility Careers
Help | Advanced Search
Computer Science > Computation and Language
Title: text generation: a systematic literature review of tasks, evaluation, and challenges.
Abstract: Text generation has become more accessible than ever, and the increasing interest in these systems, especially those using large language models, has spurred an increasing number of related publications. We provide a systematic literature review comprising 244 selected papers between 2017 and 2024. This review categorizes works in text generation into five main tasks: open-ended text generation, summarization, translation, paraphrasing, and question answering. For each task, we review their relevant characteristics, sub-tasks, and specific challenges (e.g., missing datasets for multi-document summarization, coherence in story generation, and complex reasoning for question answering). Additionally, we assess current approaches for evaluating text generation systems and ascertain problems with current metrics. Our investigation shows nine prominent challenges common to all tasks and sub-tasks in recent text generation publications: bias, reasoning, hallucinations, misuse, privacy, interpretability, transparency, datasets, and computing. We provide a detailed analysis of these challenges, their potential solutions, and which gaps still require further engagement from the community. This systematic literature review targets two main audiences: early career researchers in natural language processing looking for an overview of the field and promising research directions, as well as experienced researchers seeking a detailed view of tasks, evaluation methodologies, open challenges, and recent mitigation strategies.
Submission history
Access paper:.
- HTML (experimental)
- Other Formats
References & Citations
- Google Scholar
- Semantic Scholar
BibTeX formatted citation
Bibliographic and Citation Tools
Code, data and media associated with this article, recommenders and search tools.
- Institution
arXivLabs: experimental projects with community collaborators
arXivLabs is a framework that allows collaborators to develop and share new arXiv features directly on our website.
Both individuals and organizations that work with arXivLabs have embraced and accepted our values of openness, community, excellence, and user data privacy. arXiv is committed to these values and only works with partners that adhere to them.
Have an idea for a project that will add value for arXiv's community? Learn more about arXivLabs .
- Systematic Review
- Open access
- Published: 23 May 2024
Systematic literature review of real-world evidence for treatments in HR+/HER2- second-line LABC/mBC after first-line treatment with CDK4/6i
- Veronique Lambert ORCID: orcid.org/0000-0002-6984-0038 1 ,
- Sarah Kane ORCID: orcid.org/0009-0006-9341-4836 2 na1 ,
- Belal Howidi ORCID: orcid.org/0000-0002-1166-7631 2 na1 ,
- Bao-Ngoc Nguyen ORCID: orcid.org/0000-0001-6026-2270 2 na1 ,
- David Chandiwana ORCID: orcid.org/0009-0002-3499-2565 3 ,
- Yan Wu ORCID: orcid.org/0009-0008-3348-9232 1 ,
- Michelle Edwards ORCID: orcid.org/0009-0001-4292-3140 3 &
- Imtiaz A. Samjoo ORCID: orcid.org/0000-0003-1415-8055 2 na1
BMC Cancer volume 24 , Article number: 631 ( 2024 ) Cite this article
320 Accesses
1 Altmetric
Metrics details
Cyclin-dependent kinase 4 and 6 inhibitors (CDK4/6i) combined with endocrine therapy (ET) are currently recommended by the National Comprehensive Cancer Network (NCCN) guidelines and the European Society for Medical Oncology (ESMO) guidelines as the first-line (1 L) treatment for patients with hormone receptor-positive, human epidermal growth factor receptor 2-negative, locally advanced/metastatic breast cancer (HR+/HER2- LABC/mBC). Although there are many treatment options, there is no clear standard of care for patients following 1 L CDK4/6i. Understanding the real-world effectiveness of subsequent therapies may help to identify an unmet need in this patient population. This systematic literature review qualitatively synthesized effectiveness and safety outcomes for treatments received in the real-world setting after 1 L CDK4/6i therapy in patients with HR+/ HER2- LABC/mBC.
MEDLINE®, Embase, and Cochrane were searched using the Ovid® platform for real-world evidence studies published between 2015 and 2022. Grey literature was searched to identify relevant conference abstracts published from 2019 to 2022. The review was conducted in accordance with PRISMA guidelines (PROSPERO registration: CRD42023383914). Data were qualitatively synthesized and weighted average median real-world progression-free survival (rwPFS) was calculated for NCCN/ESMO-recommended post-1 L CDK4/6i treatment regimens.
Twenty records (9 full-text articles and 11 conference abstracts) encompassing 18 unique studies met the eligibility criteria and reported outcomes for second-line (2 L) treatments after 1 L CDK4/6i; no studies reported disaggregated outcomes in the third-line setting or beyond. Sixteen studies included NCCN/ESMO guideline-recommended treatments with the majority evaluating endocrine-based therapy; five studies on single-agent ET, six studies on mammalian target of rapamycin inhibitors (mTORi) ± ET, and three studies with a mix of ET and/or mTORi. Chemotherapy outcomes were reported in 11 studies. The most assessed outcome was median rwPFS; the weighted average median rwPFS was calculated as 3.9 months (3.3-6.0 months) for single-agent ET, 3.6 months (2.5–4.9 months) for mTORi ± ET, 3.7 months for a mix of ET and/or mTORi (3.0–4.0 months), and 6.1 months (3.7–9.7 months) for chemotherapy. Very few studies reported other effectiveness outcomes and only two studies reported safety outcomes. Most studies had heterogeneity in patient- and disease-related characteristics.
Conclusions
The real-world effectiveness of current 2 L treatments post-1 L CDK4/6i are suboptimal, highlighting an unmet need for this patient population.
Peer Review reports
Introduction
Breast cancer (BC) is the most diagnosed form of cancer in women with an estimated 2.3 million new cases diagnosed worldwide each year [ 1 ]. BC is the second leading cause of cancer death, accounting for 685,000 deaths worldwide per year [ 2 ]. By 2040, the global burden associated with BC is expected to surpass three million new cases and one million deaths annually (due to population growth and aging) [ 3 ]. Numerous factors contribute to global disparities in BC-related mortality rates, including delayed diagnosis, resulting in a high number of BC cases that have progressed to locally advanced BC (LABC) or metastatic BC (mBC) [ 4 , 5 , 6 ]. In the United States (US), the five-year survival rate for patients who progress to mBC is three times lower (31%) than the overall five-year survival rate for all stages (91%) [ 6 , 7 ].
Hormone receptor (HR) positive (i.e., estrogen receptor and/or progesterone receptor positive) coupled with negative human epidermal growth factor 2 (HER2) expression is the most common subtype of BC, accounting for ∼ 60–70% of all BC cases [ 8 , 9 ]. Historically, endocrine therapy (ET) through estrogen receptor modulation and/or estrogen deprivation has been the standard of care for first-line (1 L) treatment of HR-positive/HER2-negative (HR+/HER2-) mBC [ 10 ]. However, with the approval of the cyclin-dependent kinase 4/6 inhibitor (CDK4/6i) palbociclib in combination with the aromatase inhibitor (AI) letrozole in 2015 by the US Food and Drug Administration (FDA), 1 L treatment practice patterns have evolved such that CDK4/6i (either in combination with AIs or with fulvestrant) are currently considered the standard of care [ 11 , 12 , 13 , 14 , 15 , 16 , 17 ]. Other CDK4/6i (ribociclib and abemaciclib) in combination with ET are approved for the treatment of HR+/HER2- LABC/mBC; 1 L use of ribociclib in combination with an AI was granted FDA approval in March 2017 for postmenopausal women (with expanded approval in July 2018 for pre/perimenopausal women and for use in 1 L with fulvestrant for patients with disease progression on ET as well as for postmenopausal women), and abemaciclib in combination with fulvestrant was granted FDA approval in September 2017 for patients with disease progression following ET and as monotherapy in cases where disease progression occurs following ET and prior chemotherapy in mBC (with expanded approval in February 2018 for use in 1 L in combination with an AI for postmenopausal women) [ 18 , 19 , 20 , 21 ].
Clinical trials investigating the addition of CDK4/6i to ET have demonstrated significant improvement in progression-free survival (PFS) and significant (ribociclib) or numerical (palbociclib and abemaciclib) improvement in overall survival (OS) compared to ET alone in patients with HR+/HER2- advanced or mBC, making this combination treatment the recommended option in the 1 L setting [ 22 , 23 , 24 , 25 , 26 , 27 ]. However, disease progression occurs in a significant portion of patients after 1 L CDK4/6i treatment [ 28 ] and the optimal treatment sequence after progression on CDK4/6i remains unclear [ 29 ]. At the time of this review (literature search conducted December 14, 2022), guidelines by the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO) recommend various options for the treatment of HR+/HER2- advanced BC in the second-line (2 L) setting, including fulvestrant monotherapy, mammalian target of rapamycin inhibitors (mTORi; e.g., everolimus) ± ET, alpelisib + fulvestrant (if phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit alpha mutation positive [PIK3CA-m+]), poly-ADP ribose polymerase inhibitors (PARPi) including olaparib or talazoparib (if breast cancer gene/partner and localizer of BRCA2 positive [BRCA/PALB2m+]), and chemotherapy (in cases when a visceral crisis is present) [ 15 , 16 ]. CDK4/6i can also be used in 2 L [ 16 , 30 ]; however, limited data are available to support CDK4/6i rechallenge after its use in the 1 L setting [ 15 ]. Depending on treatments used in the 1 L and 2 L settings, treatment in the third-line setting is individualized based on the patient’s response to prior treatments, tumor load, duration of response, and patient preference [ 9 , 15 ]. Understanding subsequent treatments after 1 L CDK4/6i, and their associated effectiveness, is an important focus in BC research.
Treatment options for HR+/HER2- LABC/mBC continue to evolve, with ongoing research in both clinical trials and in the real-world setting. Real-world evidence (RWE) offers important insights into novel therapeutic regimens and the effectiveness of treatments for HR+/HER2- LABC/mBC. The effectiveness of the current treatment options following 1 L CDK4/6i therapy in the real-world setting highlights the unmet need in this patient population and may help to drive further research and drug development. In this study, we conducted a systematic literature review (SLR) to qualitatively summarize the effectiveness and safety of treatment regimens in the real-world setting after 1 L treatment with CDK4/6i in patients with HR+/HER2- LABC/mBC.
Literature search
An SLR was performed in accordance with the Cochrane Handbook for Systematic Reviews of Interventions [ 31 ] and reported in alignment with the Preferred Reporting Items for Systematic Literature Reviews and Meta-Analyses (PRISMA) statement [ 32 ] to identify all RWE studies assessing the effectiveness and safety of treatments used for patients with HR+/HER2- LABC/mBC following 1 L CDK4/6i therapy and received subsequent treatment in 2 L and beyond (2 L+). The Ovid® platform was used to search MEDLINE® (including Epub Ahead of Print and In-Process, In-Data-Review & Other Non-Indexed Citations), Ovid MEDLINE® Daily, Embase, Cochrane Central Register of Controlled Trials, and Cochrane Database of Systematic Reviews by an experienced medical information specialist. The MEDLINE® search strategy was peer-reviewed independently by a senior medical information specialist before execution using the Peer Review of Electronic Search Strategies (PRESS) checklist [ 33 ]. Searches were conducted on December 14, 2022. The review protocol was developed a priori and registered with the International Prospective Register of Systematic Review (PROSPERO; CRD42023383914) which outlined the population, intervention, comparator, outcome, and study design (PICOS) criteria and methodology used to conduct the review (Table 1 ).
Search strategies utilized a combination of controlled vocabulary (e.g., “HER2 Breast Cancer” or “HR Breast Cancer”) and keywords (e.g., “Retrospective studies”). Vocabulary and syntax were adjusted across databases. Published and validated filters were used to select for study design and were supplemented using additional medical subject headings (MeSH) terms and keywords to select for RWE and nonrandomized studies [ 34 ]. No language restrictions were included in the search strategy. Animal-only and opinion pieces were removed from the results. The search was limited to studies published between January 2015 and December 2022 to reflect the time at which FDA approval was granted for the first CDK4/6i agent (palbociclib) in combination with AI for the treatment of LABC/mBC [ 35 ]. Further search details are presented in Supplementary Material 1 .
Grey literature sources were also searched to identify relevant abstracts and posters published from January 2019 to December 2022 for prespecified relevant conferences including ESMO, San Antonio Breast Cancer Symposium (SABCS), American Society of Clinical Oncology (ASCO), the International Society for Pharmacoeconomics and Outcomes Research (ISPOR US), and the American Association for Cancer Research (AACR). A search of ClinicalTrials.gov was conducted to validate the findings from the database and grey literature searches.
Study selection, data extraction & weighted average calculation
Studies were screened for inclusion using DistillerSR Version 2.35 and 2.41 (DistillerSR Inc. 2021, Ottawa, Canada) by two independent reviewers based on the prespecified PICOS criteria (Table 1 ). A third reviewer was consulted to resolve any discrepancies during the screening process. Studies were included if they reported RWE on patients aged ≥ 18 years with HR+/HER2- LABC/mBC who received 1 L CDK4/6i treatment and received subsequent treatment in 2 L+. Studies were excluded if they reported the results of clinical trials (i.e., non-RWE), were published in any language other than English, and/or were published prior to 2015 (or prior to 2019 for conference abstracts and posters). For studies that met the eligibility criteria, data relating to study design and methodology, details of interventions, patient eligibility criteria and baseline characteristics, and outcome measures such as efficacy, safety, tolerability, and patient-reported outcomes (PROs), were extracted (as available) using a Microsoft Excel®-based data extraction form (Microsoft Corporation, WA, USA). Data extraction was performed by a single reviewer and was confirmed by a second reviewer. Multiple publications identified for the same RWE study, patient population, and setting that reported data for the same intervention were linked and extracted as a single publication. Weighted average median real-world progression-free survival (rwPFS) values were calculated by considering the contribution to the median rwPFS of each study proportional to its respective sample size. These weighted values were then used to compute the overall median rwPFS estimate.
Quality assessment
The Newcastle-Ottawa scale (NOS) for nonrandomized (cohort) studies was used to assess the risk of bias for published, full-text studies [ 36 ]. The NOS allocates a maximum of nine points for the least risk of bias across three domains: (1) Formation of study groups (four points), (2) Comparability between study groups (two points), (3) Outcome ascertainment (three points). NOS scores can be categorized in three groups: very high risk of bias (0 to 3 points), high risk of bias (4 to 6), and low risk of bias (7 to 9) [ 37 ]. Risk of bias assessment was performed by one reviewer and validated by a second independent reviewer to verify accuracy. Due to limited methodological data by which to assess study quality, risk of bias assessment was not performed on conference abstracts or posters. An amendment to the PROSPERO record (CRD42023383914) for this study was submitted in relation to the quality assessment method (specifying usage of the NOS).
The database search identified 3,377 records; after removal of duplicates, 2,759 were screened at the title and abstract stage of which 2,553 were excluded. Out of the 206 reports retrieved and assessed for eligibility, an additional 187 records were excluded after full-text review; most of these studies were excluded for having patients with mixed lines of CDK4/6i treatment (i.e., did not receive CDK4/6i exclusively in 1 L) (Fig. 1 and Table S1 ). The grey literature search identified 753 records which were assessed for eligibility; of which 752 were excluded mainly due to the population not meeting the eligibility criteria (Fig. 1 ). In total, the literature searches identified 20 records (9 published full-text articles and 11 conference abstracts/posters) representing 18 unique RWE studies that met the inclusion criteria. The NOS quality scores for the included full-text articles are provided in Table S2 . The scores ranged from four to six points (out of a total score of nine) and the median score was five, indicating that all the studies suffered from a high risk of bias [ 37 ].
Most studies were retrospective analyses of chart reviews or medical registries, and all studies were published between 2017 and 2022 (Table S3 ). Nearly half of the RWE studies (8 out of 18 studies) were conducted in the US [ 38 , 39 , 40 , 41 , 42 , 43 , 44 , 45 ], while the remaining studies included sites in Canada, China, Germany, Italy, Japan, and the United Kingdom [ 46 , 47 , 48 , 49 , 50 , 51 , 52 , 53 , 54 ]. Sample sizes ranged from as few as 4 to as many as 839 patients across included studies, with patient age ranging from 26 to 86 years old.
Although treatment characteristics in the 1 L setting were not the focus of the present review, these details are captured in Table S3 . Briefly, several RWE studies reported 1 L CDK4/6i use in combination with ET (8 out of 18 studies) or as monotherapy (2 out of 18 studies) (Table S3 ). Treatments used in combination with 1 L CDK4/6i included letrozole, fulvestrant, exemestane, and anastrozole. Where reported (4 out of 18 studies), palbociclib was the most common 1 L CDK4/6i treatment. Many studies (8 out of 18 studies) did not report which specific CDK4/6i treatment(s) were used in 1 L or if its administration was in combination or monotherapy.
Characteristics of treatments after 1 L CDK4/6i therapy
Across all studies included in this review, effectiveness and safety data were only available for treatments administered in the 2 L setting after 1 L CDK4/6i treatment. No studies were identified that reported outcomes for patients treated in the third-line setting or beyond after 1 L CDK4/6i treatment. All 18 studies reported effectiveness outcomes in 2 L, with only two of these studies also describing 2 L safety outcomes. The distribution of outcomes reported in these studies is provided in Table S4 . Studies varied in their reporting of outcomes for 2 L treatments; some studies reported outcomes for a group of 2 L treatments while others described independent outcomes for specific 2 L treatments (i.e., everolimus, fulvestrant, or chemotherapy agents such as eribulin mesylate) [ 42 , 45 , 50 , 54 , 55 ]. Due to the heterogeneity in treatment classes reported in these studies, this data was categorized (as described below) to align with the guidelines provided by NCCN and ESMO [ 15 , 16 ]. The treatment class categorizations for the purpose of this review are: single-agent ET (patients who exclusively received a single-agent ET after 1 L CDK4/6i treatment), mTORi ± ET (patients who exclusively received an mTORi with or without ET after 1 L CDK4/6i treatment), mix of ET and/or mTORi (patients who may have received only ET, only mTORi, and/or both treatments but the studies in this group lacked sufficient information to categorize these patients in the “single-agent ET” or “mTOR ± ET” categories), and chemotherapy (patients who exclusively received chemotherapy after 1 L CDK4/6i treatment). Despite ESMO and NCCN guidelines indicating that limited evidence exists to support rechallenge with CDK4/6i after 1 L CDK4/6i treatment [ 15 , 16 ], two studies reported outcomes for this treatment approach. Data for such patients were categorized as “ CDK4/6i ± ET ” as it was unclear how many patients receiving CDK4/6i rechallenge received concurrent ET. All other patient groups that lacked sufficient information or did not report outcome/safety data independently (i.e., grouped patients with mixed treatments) to categorize as one of the treatment classes described above were grouped as “ other ”.
The majority of studies reported effectiveness outcomes for endocrine-based therapy after 1 L CDK4/6i treatment; five studies for single-agent ET, six studies for mTORi ± ET, and three studies for a mix of ET and/or mTORi (Fig. 2 ). Eleven studies reported effectiveness outcomes for chemotherapy after 1 L CDK4/6i treatment, and only two studies reported effectiveness outcomes for CDK4/6i rechallenge ± ET. Eight studies that described effectiveness outcomes were grouped into the “other” category. Safety data was only reported in two studies: one study evaluating the chemotherapy agent eribulin mesylate and one evaluating the mTORi everolimus.
Effectiveness outcomes
Real-world progression-free survival
Median rwPFS was described in 13 studies (Tables 2 and Table S5 ). Across the 13 studies, the median rwPFS ranged from 2.5 months [ 49 ] to 17.3 months [ 39 ]. Out of the 13 studies reporting median rwPFS, 10 studies reported median rwPFS for a 2 L treatment recommended by ESMO and NCCN guidelines, which ranged from 2.5 months [ 49 ] to 9.7 months [ 45 ].
Weighted average median rwPFS was calculated for 2 L treatments recommended by both ESMO and NCCN guidelines (Fig. 3 ). The weighted average median rwPFS for single-agent ET was 3.9 months ( n = 92 total patients) and was derived using data from two studies reporting median rwPFS values of 3.3 months ( n = 70) [ 38 ] and 6.0 months ( n = 22) [ 40 ]. For one study ( n = 7) that reported outcomes for single agent ET, median rwPFS was not reached during the follow-up period; as such, this study was excluded from the weighted average median rwPFS calculation [ 49 ].
The weighted average median rwPFS for mTORi ± ET was 3.6 months ( n = 128 total patients) and was derived based on data from 3 studies with median rwPFS ranging from 2.5 months ( n = 4) [ 49 ] to 4.9 months ( n = 25) [ 54 ] (Fig. 3 ). For patients who received a mix of ET and/or mTORi but could not be classified into the single-agent ET or mTORi ± ET treatment classes, the weighted average median rwPFS was calculated to be 3.7 months ( n = 17 total patients). This was calculated based on data from two studies reporting median rwPFS values of 3.0 months ( n = 5) [ 46 ] and 4.0 months ( n = 12) [ 49 ]. Notably, one study of patients receiving ET and/or everolimus reported a median rwPFS duration of 3.0 months; however, this study was excluded from the weighted average median rwPFS calculation for the ET and/or mTORi class as the sample size was not reported [ 53 ].
The weighted average median rwPFS for chemotherapy was 6.1 months ( n = 499 total patients), calculated using data from 7 studies reporting median rwPFS values ranging from 3.7 months ( n = 249) [ 38 ] to 9.7 months ( n = 121) [ 45 ] (Fig. 3 ). One study with a median rwPFS duration of 5.6 months was not included in the weighted average median rwPFS calculation as the study did not report the sample size [ 53 ]. A second study was excluded from the calculation since the reported median rwPFS was not reached during the study period ( n = 7) [ 41 ].
Although 2 L CDK4/6i ± ET rechallenge lacks sufficient information to support recommendation by ESMO and NCCN guidelines, the limited data currently available for this treatment have shown promising results. Briefly, two studies reported median rwPFS for CDK4/6i ± ET with values of 8.3 months ( n = 302) [ 38 ] and 17.3 months ( n = 165) (Table 2 ) [ 39 ]. The remaining median rwPFS studies reported data for patients classified as “Other” (Table S5 ). The “Other” category included median rwPFS outcomes from seven studies, and included a myriad of treatments (e.g., ET, mTOR + ET, chemotherapy, CDK4/6i + ET, alpelisib + fulvestrant, chidamide + ET) for which disaggregated median rwPFS values were not reported.
Overall survival
Median OS for 2 L treatment was reported in only three studies (Table 2 ) [ 38 , 42 , 43 ]. Across the three studies, the 2 L median OS ranged from 5.2 months ( n = 3) [ 43 ] to 35.7 months ( n = 302) [ 38 ]. Due to the lack of OS data in most of the studies, weighted averages could not be calculated. No median OS data was reported for the single-agent ET treatment class whereas two studies reported median OS for the mTORi ± ET treatment class, ranging from 5.2 months ( n = 3) [ 43 ] to 21.8 months ( n = 54) [ 42 ]. One study reported 2 L median OS of 24.8 months for a single patient treated with chemotherapy [ 43 ]. The median OS data in the CDK4/6i ± ET rechallenge group was 35.7 months ( n = 302) [ 38 ].
Patient mortality was reported in three studies [ 43 , 44 , 45 ]. No studies reported mortality for the single-agent ET treatment class and only one study reported this outcome for the mTORi ± ET treatment class, where 100% of patients died ( n = 3) as a result of rapid disease progression [ 43 ]. For the chemotherapy class, one study reported mortality for one patient receiving 2 L capecitabine [ 43 ]. An additional study reported eight deaths (21.7%) following 1 L CDK4/6i treatment; however, this study did not disclose the 2 L treatments administered to these patients [ 44 ].
Other clinical endpoints
The studies included limited information on additional clinical endpoints; two studies reported on time-to-discontinuation (TTD), two reported on duration of response (DOR), and one each on time-to-next-treatment (TTNT), time-to-progression (TTP), objective response rate (ORR), clinical benefit rate (CBR), and stable disease (Tables 2 and Table S5 ).
Safety, tolerability, and patient-reported outcomes
Safety and tolerability data were reported in two studies [ 40 , 45 ]. One study investigating 2 L administration of the chemotherapy agent eribulin mesylate reported 27 patients (22.3%) with neutropenia, 3 patients (2.5%) with febrile neutropenia, 10 patients (8.3%) with peripheral neuropathy, and 14 patients (11.6%) with diarrhea [ 45 ]. Of these, neutropenia of grade 3–4 severity occurred in 9 patients (33.3%) [ 45 ]. A total of 55 patients (45.5%) discontinued eribulin mesylate treatment; 1 patient (0.83%) discontinued treatment due to adverse events [ 45 ]. Another study reported that 5 out of the 22 patients receiving the mTORi everolimus combined with ET in 2 L (22.7%) discontinued treatment due to toxicity [ 40 ]. PROs were not reported in any of the studies included in the SLR.
The objective of this study was to summarize the existing RWE on the effectiveness and safety of therapies for patients with HR+/HER2- LABC/mBC after 1 L CDK4/6i treatment. We identified 18 unique studies reporting specifically on 2 L treatment regimens after 1 L CDK4/6i treatment. The weighted average median rwPFS for NCCN- and ESMO- guideline recommended 2 L treatments ranged from 3.6 to 3.9 months for ET-based treatments and was 6.1 months when including chemotherapy-based regimens. Treatment selection following 1 L CDK4/6i therapy remains challenging primarily due to the suboptimal effectiveness or significant toxicities (e.g., chemotherapy) associated with currently available options [ 56 ]. These results highlight that currently available 2 L treatments for patients with HR+/HER2- LABC/mBC who have received 1 L CDK4/6i are suboptimal, as evidenced by the brief median rwPFS duration associated with ET-based treatments, or notable side effects and toxicity linked to chemotherapy. This conclusion is aligned with a recent review highlighting the limited effectiveness of treatment options for HR+/HER2- LABC/mBC patients post-CDK4/6i treatment [ 56 , 57 ]. Registrational trials which have also shed light on the short median PFS of 2–3 months achieved by ET (i.e., fulvestrant) after 1 L CDK4/6i therapy emphasize the need to develop improved treatment strategies aimed at prolonging the duration of effective ET-based treatment [ 56 ].
The results of this review reveal a paucity of additional real-world effectiveness and safety evidence after 1 L CDK4/6i treatment in HR+/HER2- LABC/mBC. OS and DOR were only reported in two studies while other clinical endpoints (i.e., TTD, TTNT, TTP, ORR, CBR, and stable disease) were only reported in one study each. Similarly, safety and tolerability data were only reported in two studies each, and PROs were not reported in any study. This hindered our ability to provide a comprehensive assessment of real-world treatment effectiveness and safety following 1 L CDK4/6i treatment. The limited evidence may be due to the relatively short period of time that has elapsed since CDK4/6i first received US FDA approval for 1 L treatment of HR+/HER2- LABC/mBC (2015) [ 35 ]. As such, almost half of our evidence was informed by conference abstracts. Similarly, no real-world studies were identified in our review that reported outcomes for treatments in the third- or later-lines of therapy after 1 L CDK4/6i treatment. The lack of data in this patient population highlights a significant gap which limits our understanding of the effectiveness and safety for patients receiving later lines of therapy. As more patients receive CDK4/6i therapy in the 1 L setting, the number of patients requiring subsequent lines of therapy will continue to grow. Addressing this data gap over time will be critical to improve outcomes for patients with HR+/HER2- LABC/mBC following 1 L CDK4/6i therapy.
There are several strengths of this study, including adherence to the guidelines outlined in the Cochrane Handbook to ensure a standardized and reliable approach to the SLR [ 58 ] and reporting of the SLR following PRISMA guidelines to ensure transparency and reproducibility [ 59 ]. Furthermore, the inclusion of only RWE studies allowed us to assess the effectiveness of current standard of care treatments outside of a controlled environment and enabled us to identify an unmet need in this patient population.
This study had some notable limitations, including the lack of safety and additional effectiveness outcomes reported. In addition, the dearth of studies reporting PROs is a limitation, as PROs provide valuable insight into the patient experience and are an important aspect of assessing the impact of 2 L treatments on patients’ quality of life. The studies included in this review also lacked consistent reporting of clinical characteristics (e.g., menopausal status, sites of metastasis, prior surgery) making it challenging to draw comprehensive conclusions or comparisons based on these factors across the studies. Taken together, there exists an important gap in our understanding of the long-term management of patients with HR+/HER2- LABC/mBC. Additionally, the effectiveness results reported in our evidence base were informed by small sample sizes; many of the included studies reported median rwPFS based on less than 30 patients [ 39 , 40 , 41 , 46 , 49 , 51 , 60 ], with two studies not reporting the sample size at all [ 47 , 53 ]. This may impact the generalizability and robustness of the results. Relatedly, the SLR database search was conducted in December 2022; as such, novel agents (e.g., elacestrant and capivasertib + fulvestrant) that have since received FDA approval for the treatment of HR+/HER2- LABC/mBC may impact current 2 L rwPFS outcomes [ 61 , 62 ]. Finally, relative to the number of peer-reviewed full-text articles, this SLR identified eight abstracts and one poster presentation, comprising half (50%) of the included unique studies. As conference abstracts are inherently limited by how much content that can be described due to word limit constraints, this likely had implications on the present synthesis whereby we identified a dearth of real-world effectiveness outcomes in patients with HR+/HER2- LABC/mBC treated with 1 L CDK4/6i therapy.
Future research in this area should aim to address the limitations of the current literature and provide a more comprehensive understanding of optimal sequencing of effective and safe treatment for patients following 1 L CDK4/6i therapy. Specifically, future studies should strive to report robust data related to effectiveness, safety, and PROs for patients receiving 2 L treatment after 1 L CDK4/6i therapy. Future studies should also aim to understand the mechanism underlying CDK4/6i resistance. Addressing these gaps in knowledge may improve the long-term real-world management of patients with HR+/HER2- LABC/mBC. A future update of this synthesis may serve to capture a wider breadth of full-text, peer-reviewed articles to gain a more robust understanding of the safety, effectiveness, and real-world treatment patterns for patients with HR+/HER2- LABC/mBC. This SLR underscores the necessity for ongoing investigation and the development of innovative therapeutic approaches to address these gaps and improve patient outcomes.
This SLR qualitatively summarized the existing real-world effectiveness data for patients with HR+/HER2- LABC/mBC after 1 L CDK4/6i treatment. Results of this study highlight the limited available data and the suboptimal effectiveness of treatments employed in the 2 L setting and underscore the unmet need in this patient population. Additional studies reporting effectiveness and safety outcomes, in addition to PROs, for this patient population are necessary and should be the focus of future research.
PRISMA flow diagram. *Two included conference abstracts reported the same information as already included full-text reports, hence both conference abstracts were not identified as unique. Abbreviations: 1 L = first-line; AACR = American Association of Cancer Research; ASCO = American Society of Clinical Oncology; CDK4/6i = cyclin-dependent kinase 4/6 inhibitor; ESMO = European Society for Medical Oncology; ISPOR = Professional Society for Health Economics and Outcomes Research; n = number of studies; NMA = network meta-analysis; pts = participants; SABCS = San Antonio Breast Cancer Symposium; SLR = systematic literature review.
Number of studies reporting effectiveness outcomes exclusively for each treatment class. *Studies that lack sufficient information on effectiveness outcomes to classify based on the treatment classes outlined in the legend above. Abbreviations: CDK4/6i = cyclin-dependent kinase 4/6 inhibitor; ET = endocrine therapy; mTORi = mammalian target of rapamycin inhibitor.
Weighted average median rwPFS for 2 L treatments (recommended in ESMO/NCCN guidelines) after 1 L CDK4/6i treatment. Circular dot represents weighted average median across studies. Horizontal bars represent the range of values reported in these studies. Abbreviations: CDK4/6i = cyclin-dependent kinase 4/6 inhibitor; ESMO = European Society for Medical Oncology; ET = endocrine therapy, mTORi = mammalian target of rapamycin inhibitor; n = number of patients; NCCN = National Comprehensive Cancer Network; rwPFS = real-world progression-free survival.
Data availability
All data generated or analyzed during this study are included in this published article [and its supplementary information files]. This study is registered with PROSPERO (CRD42023383914).
Abbreviations
Second-line
Second-line treatment setting and beyond
American Association of Cancer Research
Aromatase inhibitor
American Society of Clinical Oncology
- Breast cancer
breast cancer gene/partner and localizer of BRCA2 positive
Clinical benefit rate
Cyclin-dependent kinase 4/6 inhibitor
Complete response
Duration of response
European Society for Medical Oncology
Food and Drug Administration
Human epidermal growth factor receptor 2
Human epidermal growth factor receptor 2 negative
Hormone receptor
Hormone receptor positive
Professional Society for Health Economics and Outcomes Research
Locally advanced breast cancer
Metastatic breast cancer
Medical Literature Analysis and Retrieval System Online
Medical subject headings
Mammalian target of rapamycin inhibitor
National Comprehensive Cancer Network
Newcastle Ottawa Scale
Objective response rate
Poly-ADP ribose polymerase inhibitor
Progression-free survival
Population, Intervention, Comparator, Outcome, Study Design
Partial response
Preferred Reporting Items for Systematic Literature Reviews and Meta-Analyses
Patient-reported outcomes
- Real-world evidence
San Antonio Breast Cancer Symposium
- Systematic literature review
Time-to-discontinuation
Time-to-next-treatment
Time-to-progression
United States
Łukasiewicz S, Czeczelewski M, Forma A, Baj J, Sitarz R, Stanisławek A, Breast, Cancer—Epidemiology. Risk factors, classification, prognostic markers, and current treatment Strategies—An. Updated Rev Cancers. 2021;13(17):4287.
Google Scholar
World Health Organization (WHO). Breast Cancer Facts Sheet [updated July 12 2023. https://www.who.int/news-room/fact-sheets/detail/breast-cancer .
Arnold M, Morgan E, Rumgay H, Mafra A, Singh D, Laversanne M, et al. Current and future burden of breast cancer: global statistics for 2020 and 2040. Breast. 2022;66:15–23.
Article PubMed PubMed Central Google Scholar
Wilkinson L, Gathani T. Understanding breast cancer as a global health concern. Br J Radiol. 2022;95(1130):20211033.
Article PubMed Google Scholar
Giaquinto AN, Sung H, Miller KD, Kramer JL, Newman LA, Minihan A et al. Breast Cancer Statistics, 2022. CA: A Cancer Journal for Clinicians. 2022;72(6):524– 41.
National Cancer Institute (NIH). Cancer Stat Facts: Female Breast Cancer [updated 2020. https://seer.cancer.gov/statfacts/html/breast.html .
American Cancer Society. Key Statistics for Breast Cancer [ https://www.cancer.org/cancer/types/breast-cancer/about/how-common-is-breast-cancer.html .
Zagami P, Carey LA. Triple negative breast cancer: pitfalls and progress. npj Breast Cancer. 2022;8(1):95.
Article CAS PubMed PubMed Central Google Scholar
Matutino A, Joy AA, Brezden-Masley C, Chia S, Verma S. Hormone receptor-positive, HER2-negative metastatic breast cancer: redrawing the lines. Curr Oncol. 2018;25(Suppl 1):S131–41.
Lloyd MR, Wander SA, Hamilton E, Razavi P, Bardia A. Next-generation selective estrogen receptor degraders and other novel endocrine therapies for management of metastatic hormone receptor-positive breast cancer: current and emerging role. Ther Adv Med Oncol. 2022;14:17588359221113694.
Cardoso F, Senkus E, Costa A, Papadopoulos E, Aapro M, André F, et al. 4th ESO-ESMO International Consensus guidelines for advanced breast Cancer (ABC 4)†. Ann Oncol. 2018;29(8):1634–57.
Article CAS PubMed Google Scholar
US Food Drug Administration. Palbociclib (Ibrance) 2017 [updated March 31, 2017. https://www.fda.gov/drugs/resources-information-approved-drugs/palbociclib-ibrance .
US Food Drug Administration. FDA expands ribociclib indication in HR-positive, HER2-negative advanced or metastatic breast cancer 2018 [updated July 18. 2018. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-expands-ribociclib-indication-hr-positive-her2-negative-advanced-or-metastatic-breast-cancer .
US Food Drug Administration. FDA approves abemaciclib for HR positive, HER2-negative breast cancer 2017 [updated Sept 28. 2017. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-abemaciclib-hr-positive-her2-negative-breast-cancer .
NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®). Breast Cancer 2022 [ https://www.nccn.org/professionals/physician_gls/pdf/breast.pdf .
Gennari A, André F, Barrios CH, Cortés J, de Azambuja E, DeMichele A, et al. ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer. Ann Oncol. 2021;32(12):1475–95.
Beaver JA, Amiri-Kordestani L, Charlab R, Chen W, Palmby T, Tilley A, et al. FDA approval: Palbociclib for the Treatment of Postmenopausal Patients with estrogen Receptor-Positive, HER2-Negative metastatic breast Cancer. Clin Cancer Res. 2015;21(21):4760–6.
US Food Drug Administration. Ribociclib (Kisqali) [ https://www.fda.gov/drugs/resources-information-approved-drugs/ribociclib-kisqali#:~:text=On%20March%2013%2C%202017%2C%20the,hormone%20receptor%20(HR)%2Dpositive%2C .
US Food Drug Administration. FDA approves new treatment for certain advanced or metastatic breast cancers [ https://www.fda.gov/news-events/press-announcements/fda-approves-new-treatment-certain-advanced-or-metastatic-breast-cancers .
US Food Drug Administration. FDA expands ribociclib indication in HR-positive, HER2-negative advanced or metastatic breast cancer. 2018 [ https://www.fda.gov/drugs/resources-information-approved-drugs/fda-expands-ribociclib-indication-hr-positive-her2-negative-advanced-or-metastatic-breast-cancer .
US Food Drug Administration. FDA approves abemaciclib as initial therapy for HR-positive, HER2-negative metastatic breast cancer [ https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-abemaciclib-initial-therapy-hr-positive-her2-negative-metastatic-breast-cancer .
Turner NC, Slamon DJ, Ro J, Bondarenko I, Im S-A, Masuda N, et al. Overall survival with Palbociclib and fulvestrant in advanced breast Cancer. N Engl J Med. 2018;379(20):1926–36.
Slamon DJ, Neven P, Chia S, Fasching PA, De Laurentiis M, Im SA, et al. Phase III randomized study of Ribociclib and Fulvestrant in hormone Receptor-Positive, human epidermal growth factor receptor 2-Negative advanced breast Cancer: MONALEESA-3. J Clin Oncol. 2018;36(24):2465–72.
Goetz MP, Toi M, Campone M, Sohn J, Paluch-Shimon S, Huober J, et al. MONARCH 3: Abemaciclib as initial therapy for advanced breast Cancer. J Clin Oncol. 2017;35(32):3638–46.
Gopalan PK, Villegas AG, Cao C, Pinder-Schenck M, Chiappori A, Hou W, et al. CDK4/6 inhibition stabilizes disease in patients with p16-null non-small cell lung cancer and is synergistic with mTOR inhibition. Oncotarget. 2018;9(100):37352–66.
Watt AC, Goel S. Cellular mechanisms underlying response and resistance to CDK4/6 inhibitors in the treatment of hormone receptor-positive breast cancer. Breast Cancer Res. 2022;24(1):17.
Goetz M. MONARCH 3: final overall survival results of abemaciclib plus a nonsteroidal aromatase inhibitor as first-line therapy for HR+, HER2- advanced breast cancer. SABCS; 2023.
Munzone E, Pagan E, Bagnardi V, Montagna E, Cancello G, Dellapasqua S, et al. Systematic review and meta-analysis of post-progression outcomes in ER+/HER2– metastatic breast cancer after CDK4/6 inhibitors within randomized clinical trials. ESMO Open. 2021;6(6):100332.
Gennari A, André F, Barrios CH, Cortés J, de Azambuja E, DeMichele A, et al. ESMO Clinical Practice Guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer. Annals of Oncology. 2021;32(12):1475-95.
European Society for Medical Oncology (ESMO). ESMO Metastatic Breast Cancer Living Guideline: ER-positive HER2-negative Breast Cancer [updated May 2023. https://www.esmo.org/living-guidelines/esmo-metastatic-breast-cancer-living-guideline/er-positive-her2-negative-breast-cancer .
Higgins JPT, Thomas J, Chandler J, Cumpston M, Li T, Welch PM VA, editors. Cochrane Handbook for Systematic Reviews of Interventions version 6.2 (updated February 2021). www.training.cochrane.org/handbook : Cochrane; 2021.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLoS Med. 2021;18(3):e1003583.
McGowan J, Sampson M, Salzwedel DM, Cogo E, Foerster V, Lefebvre C. PRESS peer review of electronic search strategies: 2015 Guideline Statement. J Clin Epidemiol. 2016;75:40–6.
Fraser C, Murray A, Burr J. Identifying observational studies of surgical interventions in MEDLINE and EMBASE. BMC Med Res Methodol. 2006;6(1):41.
US Food Drug Administration. Palbociclib (Ibrance). Silver Spring, MD: US Food and Drug Administration; 2017.
Book Google Scholar
GA Wells BS, D O’Connell J, Peterson V, Welch M, Losos PT. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses [ https://www.ohri.ca/programs/clinical_epidemiology/oxford.asp .
Lo CK-L, Mertz D, Loeb M. Newcastle-Ottawa Scale: comparing reviewers’ to authors’ assessments. BMC Med Res Methodol. 2014;14(1):45.
Martin JM, Handorf EA, Montero AJ, Goldstein LJ. Systemic therapies following progression on first-line CDK4/6-inhibitor treatment: analysis of real-world data. Oncologist. 2022;27(6):441–6.
Kalinsky KM, Kruse M, Smyth EN, Guimaraes CM, Gautam S, Nisbett AR et al. Abstract P1-18-37: Treatment patterns and outcomes associated with sequential and non-sequential use of CDK4 and 6i for HR+, HER2- MBC in the real world. Cancer Research. 2022;82(4_Supplement):P1-18-37-P1-18-37.
Choong GM, Liddell S, Ferre RAL, O’Sullivan CC, Ruddy KJ, Haddad TC, et al. Clinical management of metastatic hormone receptor-positive, HER2-negative breast cancer (MBC) after CDK 4/6 inhibitors: a retrospective single-institution study. Breast Cancer Res Treat. 2022;196(1):229–37.
Xi J, Oza A, Thomas S, Ademuyiwa F, Weilbaecher K, Suresh R, et al. Retrospective Analysis of Treatment Patterns and effectiveness of Palbociclib and subsequent regimens in metastatic breast Cancer. J Natl Compr Canc Netw. 2019;17(2):141–7.
Rozenblit M, Mun S, Soulos P, Adelson K, Pusztai L, Mougalian S. Patterns of treatment with everolimus exemestane in hormone receptor-positive HER2-negative metastatic breast cancer in the era of targeted therapy. Breast Cancer Res. 2021;23(1):14.
Bashour SI, Doostan I, Keyomarsi K, Valero V, Ueno NT, Brown PH, et al. Rapid breast Cancer Disease Progression following cyclin dependent kinase 4 and 6 inhibitor discontinuation. J Cancer. 2017;8(11):2004–9.
Giridhar KV, Choong GM, Leon-Ferre R, O’Sullivan CC, Ruddy K, Haddad T, et al. Abstract P6-18-09: clinical management of metastatic breast cancer (MBC) after CDK 4/6 inhibitors: a retrospective single-institution study. Cancer Res. 2019;79:P6–18.
Article Google Scholar
Mougalian SS, Feinberg BA, Wang E, Alexis K, Chatterjee D, Knoth RL, et al. Observational study of clinical outcomes of eribulin mesylate in metastatic breast cancer after cyclin-dependent kinase 4/6 inhibitor therapy. Future Oncol. 2019;15(34):3935–44.
Moscetti LML, Riggi L, Sperduti I, Piacentini FOC, Toss A, Barbieri E, Cortesi L, Canino FMA, Zoppoli G, Frassoldati A, Schirone A, Dominici MECF. SEQUENCE OF TREATMENTS AFTER CDK4/6 THERAPY IN ADVANCED BREAST CANCER (ABC), A GOIRC MULTICENTER RETRO/ PROSPECTIVE STUDY. PRELIMINARY RESULTS IN THE RETROSPECTIVE SERIES OF 116 PATIENTS. Tumori. 2022;108(4S):80.
Menichetti AZE, Giorgi CA, Bottosso M, Leporati R, Giarratano T, Barbieri C, Ligorio F, Mioranza E, Miglietta F, Lobefaro R, Faggioni G, Falci C, Vernaci G, Di Liso E, Girardi F, Griguolo G, Vernieri C, Guarneri V, Dieci MV. CDK 4/6 INHIBITORS FOR METASTATIC BREAST CANCER: A MULTICENTER REALWORLD STUDY. Tumori. 2022;108(4S):70.
Marschner NW, Harbeck N, Thill M, Stickeler E, Zaiss M, Nusch A, et al. 232P Second-line therapies of patients with early progression under CDK4/6-inhibitor in first-line– data from the registry platform OPAL. Annals of Oncology. 2022;33:S643-S4
Gousis C, Lowe KMH, Kapiris M. V. Angelis. Beyond First Line CDK4/6 Inhibitors (CDK4/6i) and Aromatase Inhibitors (AI) in Patients with Oestrogen Receptor Positive Metastatic Breast Cancer (ERD MBC): The Guy’s Cancer Centre Experience. Clinical Oncology2022. p. e178.
Endo Y, Yoshimura A, Sawaki M, Hattori M, Kotani H, Kataoka A, et al. Time to chemotherapy for patients with estrogen receptor-positive breast Cancer and cyclin-dependent kinase 4 and 6 inhibitor use. J Breast Cancer. 2022;25(4):296–306.
Li Y, Li W, Gong C, Zheng Y, Ouyang Q, Xie N, et al. A multicenter analysis of treatment patterns and clinical outcomes of subsequent therapies after progression on palbociclib in HR+/HER2- metastatic breast cancer. Ther Adv Med Oncol. 2021;13:17588359211022890.
Amaro CP, Batra A, Lupichuk S. First-line treatment with a cyclin-dependent kinase 4/6 inhibitor plus an aromatase inhibitor for metastatic breast Cancer in Alberta. Curr Oncol. 2021;28(3):2270–80.
Crocetti SPM, Tassone L, Marcantognini G, Bastianelli L, Della Mora A, Merloni F, Cantini L, Scortichini L, Agostinelli V, Ballatore Z, Savini A, Maccaroni E. Berardi R. What is the best therapeutic sequence for ER-Positive/HER2- Negative metastatic breast cancer in the era of CDK4/6 inhibitors? A single center experience. Tumori. 2020;106(2S).
Nichetti F, Marra A, Giorgi CA, Randon G, Scagnoli S, De Angelis C, et al. 337P Efficacy of everolimus plus exemestane in CDK 4/6 inhibitors-pretreated or naïve HR-positive/HER2-negative breast cancer patients: A secondary analysis of the EVERMET study. Annals of Oncology. 2020;31:S382
Luhn P, O’Hear C, Ton T, Sanglier T, Hsieh A, Oliveri D, et al. Abstract P4-13-08: time to treatment discontinuation of second-line fulvestrant monotherapy for HR+/HER2– metastatic breast cancer in the real-world setting. Cancer Res. 2019;79(4Supplement):P4–13.
Mittal A, Molto Valiente C, Tamimi F, Schlam I, Sammons S, Tolaney SM et al. Filling the gap after CDK4/6 inhibitors: Novel Endocrine and Biologic Treatment options for metastatic hormone receptor positive breast Cancer. Cancers (Basel). 2023;15(7).
Ashai N, Swain SM. Post-CDK 4/6 inhibitor therapy: current agents and novel targets. Cancers (Basel). 2023;15(6).
Higgins JPTTJ, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editors. Cochrane Handbook for Systematic Reviews of Interventions version 6.3 (updated February 2022). www.training.cochrane.org/handbook : Cochrane; 2022.
Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71.
Serdar CC, Cihan M, Yücel D, Serdar MA. Sample size, power and effect size revisited: simplified and practical approaches in pre-clinical, clinical and laboratory studies. Biochem Med (Zagreb). 2021;31(1):010502.
US Food Drug Administration. FDA approves elacestrant for ER-positive, HER2-negative, ESR1-mutated advanced or metastatic breast cancer [updated January 27 2023. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-elacestrant-er-positive-her2-negative-esr1-mutated-advanced-or-metastatic-breast-cancer .
US Food Drug Administration. FDA approves capivasertib with fulvestrant for breast cancer [updated November 16 2023. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-capivasertib-fulvestrant-breast-cancer .
Download references
Acknowledgements
The authors would like to acknowledge Joanna Bielecki who developed, conducted, and documented the database searches.
This study was funded by Pfizer Inc. (New York, NY, USA) and Arvinas (New Haven, CT, USA).
Author information
Sarah Kane, Belal Howidi, Bao-Ngoc Nguyen and Imtiaz A. Samjoo contributed equally to this work.
Authors and Affiliations
Pfizer, 10017, New York, NY, USA
Veronique Lambert & Yan Wu
EVERSANA, Burlington, ON, Canada
Sarah Kane, Belal Howidi, Bao-Ngoc Nguyen & Imtiaz A. Samjoo
Arvinas, 06511, New Haven, CT, USA
David Chandiwana & Michelle Edwards
You can also search for this author in PubMed Google Scholar
Contributions
VL, IAS, SK, BH, BN, DC, YW, and ME participated in the conception and design of the study. IAS, SK, BH and BN contributed to the literature review, data collection, analysis, and interpretation of the data. VL, IAS, SK, BH, BN, DC, YW, and ME contributed to the interpretation of the data and critically reviewed for the importance of intellectual content for the work. VL, IAS, SK, BH, BN, DC, YW, and ME were responsible for drafting or reviewing the manuscript and for providing final approval. VL, IAS, SK, BH, BN, DC, YW, and ME meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work, and have given their approval for this version to be published.
Corresponding author
Correspondence to Imtiaz A. Samjoo .
Ethics declarations
Ethics approval and consent to participate.
Not applicable.
Consent for publication
Competing interests.
The authors of this manuscript declare that the research presented was funded by Pfizer Inc. and Arvinas. While the support from Pfizer Inc. and Arvinas was instrumental in facilitating this research, the authors affirm that their interpretation of the data and the content of this manuscript were conducted independently and without bias to maintain the transparency and integrity of the research. IAS, SK, BH, and BN are employees of EVERSANA, Canada, which was a paid consultant to Pfizer in connection with the development of this manuscript.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1
Supplementary material 2, rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Lambert, V., Kane, S., Howidi, B. et al. Systematic literature review of real-world evidence for treatments in HR+/HER2- second-line LABC/mBC after first-line treatment with CDK4/6i. BMC Cancer 24 , 631 (2024). https://doi.org/10.1186/s12885-024-12269-8
Download citation
Received : 26 January 2024
Accepted : 16 April 2024
Published : 23 May 2024
DOI : https://doi.org/10.1186/s12885-024-12269-8
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- First-line CDK4/6i
ISSN: 1471-2407
- Submission enquiries: [email protected]
- General enquiries: [email protected]
Systematic Literature Review in the Field of Art and Business
- First Online: 30 August 2020
Cite this chapter

- Stefania Masè ORCID: orcid.org/0000-0002-4685-4453 17
Part of the book series: International Series in Advanced Management Studies ((ISAMS))
583 Accesses
1 Citations
As a burgeoning area of research, Arts and Business still lacks clearly defined boundaries, thus requiring the largest possible contribution from previously validated research. So as not to neglect any important contributions, the review of the relevant literature which follows focuses mainly on research conducted in the area of management. In order to assure that literature review was as complete as possible, we adopted a method called systematic literature review. After an overview of this methodology, its application will be deeply described. A description of the results obtained will follow, together with the main literature strands highlighted in the field of Art and Business.
This chapter is revised and updated from the author’s dissertation Art & Business from sponsorship and philanthropy to the contemporary process of artification , Sorbonne U. and Macerata U. completed in 2016.
This is a preview of subscription content, log in via an institution to check access.
Access this chapter
- Available as EPUB and PDF
- Read on any device
- Instant download
- Own it forever
- Compact, lightweight edition
- Dispatched in 3 to 5 business days
- Free shipping worldwide - see info
- Durable hardcover edition
Tax calculation will be finalised at checkout
Purchases are for personal use only
Institutional subscriptions
It should be noted, that Daragh O’Reilly’s study refers to the article by Rentschler and Kirchner entitled “Who is publishing and citing arts management/marketing articles? Examining the external impact of arts management journals,” published as act of the ninth International Colloquium on Arts, Heritage, Non Profit and Social Marketing conference, held in King’s College of London in September 2010. For the present study, instead, article of Rentschler and Kirchner published in 2012 on Arts Marketing: an international journal has been used. The list of the major journals in the field of arts management has not changed in the two different versions of the research.
Alexander, V. D. (2014). Art and the twenty-first century gift: Corporate philanthropy and government funding in the cultural sector. Anthropological Forum, 24 (4), 364–380. https://doi.org/10.1080/00664677.2014.947917
Article Google Scholar
Alexander, V. D. (1996). From philanthropy to funding: The effects of corporate and public support on American art museums. Poetics, 24 (2–4), 87–129. https://doi.org/10.1016/0304-422X(95)00003-3
Alfakhri, D., Harness, D., Nicholson, J., & Harness, T. (2018). The role of aesthetics and design in hotelscape: A phenomenological investigation of cosmopolitan consumers. Journal of Business Research, 85 , 523–531. https://doi.org/10.1016/j.jbusres.2017.10.031
An, D., & Youn, N. (2018). The inspirational power of arts on creativity. Journal of Business Research, 85 , 467–475. https://doi.org/10.1016/j.jbusres.2017.10.025
Areni, C. S. (2003). Examining managers’ theories of how atmospheric music affects perception, behaviour and financial performance. Journal of Retailing and Consumer Services, 10 (5), 263–274. https://doi.org/10.1016/S0969-6989(02)00063-2
Barends, E., Rousseau, D. M. & Briner, R. B. (2014). Evidence-based management: The basic principles . Amsterdam: Center for Evidence-Based Management. Accessed August 18, 2020, from https://www.cebma.org/wp-content/uploads/Evidence-Based-Practice-The-Basic-Principles.pdf
Baumgarth, C. (2018). Brand management and the world of the arts: Collaboration, co-operation, co-creation, and inspiration. Journal of Product & Brand Management, 27 (3), 237–248. https://doi.org/10.1108/JPBM-03-2018-1772
Behnke, C. (2007). Corporate art collecting: A survey of German-speaking companies. The Journal of Arts Management, Law, and Society, 37 (3), 225–244. https://doi.org/10.3200/JAML.37.3.225-244
Bloch, M. (1989). Corporate social responsibility and the arts. Journal of Arts Management and Law, 19 (2), 23–32. https://doi.org/10.1080/07335113.1989.9943118
Borghini, S., Visconti, L. M., Anderson, L., & Sherry Jr., J. F. (2010). Symbiotic postures of commercial advertising and street art. Journal of Advertising, 39 (3), 113–126. https://doi.org/10.2753/JOA0091-3367390308
Brennan, L., Binney, W., & Brady, E. (2012). The raising of corporate sponsorship: A behavioral study. Journal of Nonprofit and Public Sector Marketing, 24 (3), 222–237. https://doi.org/10.1080/10495142.2012.705181
Briner, R. B., Denyer, D. & Rousseau, D.M. (2009). Evidence-Based Management: Concept Cleanup Time? Academy of Management Perspectives (November), 19–32. https://doi.org/10.5465/amp.23.4.19
Briner, R. B., & Walshe, N. D. (2014). From passively received wisdom to actively constructed knowledge: Teaching Systematic review skills as a Foundation of Evidence-Based Management. Academy of Management Learning & Education, 13 (3), 415–432. https://doi.org/10.5465/amle.2013.0222
Bucher, V. (1989). Art and cultural sponsorship ‘Austrian -style’. The International Journal of Museum Management and Curatorship, 8 , 77–82. https://doi.org/10.1080/09647778909515153
Bulut, D., & Bulut Yumrukaka, C. (2009). Corporate social responsibility in culture and art. Management of Environmental Quality: An International Journal, 20 (3), 311–320. https://doi.org/10.1108/14777830910950702
Carlucci, D., & Schiuma, G. (2018). The power of the arts in business. Journal of Business Research, 85 , 342–347. https://doi.org/10.1016/j.jbusres.2017.10.012
Carrillat, F. A., D’astous, A., & Colbert, F. (2008). The effectiveness of art venue sponsorship: An attribution perspective. Journal of Sponsorship, 1 (3), 274–285. http://hdl.handle.net/10453/29821
Carù, A., Ostillio, M., & Leone, G. (2017). Corporate museums to enhance brand authenticity in luxury goods companies: The case of Salvatore Ferragamo. International Journal of Arts Management, 19 (2), 32–45. ISSN 1480-8986, ZDB-ID 2105595-6.
Google Scholar
Cassullo, J. L. (1988). The fine art of corporate collecting. Harvard Business Review, 66 (6), 137–141.
Chailan, C. (2018). Art as a means to recreate luxury brands’ rarity and value. Journal of Business Research . https://doi.org/10.1016/j.jbusres.2017.10.019
Chaney, D., Pulh, M., & Mencarelli, R. (2017). When arts inspire businesses: Museums as tool of brands. Journal of Business Research, 85 , 452–458. https://doi.org/10.1016/j.jbusres.2017.10.023
Choi, E., Ko, E., & Kim, A. J. (2018). Explaining and predicting purchase intentions following luxury-fashion brand value co-creation encounters. Journal of Business Research, 69 (12), 5827–5832. https://doi.org/10.1016/j.jbusres.2016.04.180
Chong, D. (2000). Re-readings in arts management. Journal of Arts Management, Law & Society, 29 (4), 290–303. https://doi.org/10.1080/10632920009597307
Cochrane EPOC Effective Practice and Organisation of Care (2017). How to develop a search strategy for an intervention review . Accessed June 15, 2020, from https://epoc.cochrane.org/sites/epoc.cochrane.org/files/public/uploads/Resources-for-authors2017/how_to_develop_a_search_strategy.pdf
Colbert, F., D’astous, A., & Parmentier, M. A. (2005a). Consumer perceptions of sponsorship in the arts. International Journal of Cultural Policy, 11 (2), 215–228. https://doi.org/10.1080/10286630500198245
Colbert, F., D’astous, A., & Parmentier, M. A. (2005b). Consumer perception of private versus public sponsorship of the arts. International Journal of Arts Management, 8 (1), 48–60. http://www.jstor.org/stable/41064862
Comunian, R. (2008). Culture Italian style: Business and the arts. Journal of Business Strategy, 29 (3), 37–44. https://doi.org/10.1108/02756660810873209
Comunian, R. (2009). Towards a new conceptual framework for business investments in the arts: Some examples from Italy. The Journal of Arts Management, Law, and Society, 39 (3), 200–220. https://doi.org/10.1080/10632920903218521
Cwi, D., & Strausbaugh, J. (1988). The royalists, the realists, and the radicals: A comparative analysis of arts funding in Canada and the United States. Journal of Cultural Economics, 12 (1). http://www.jstor.org/stable/41810396
Daellenbach, K. (2012). Understanding the decision-making processes for arts sponsorship. International Journal of Nonprofit & Voluntary Sector Marketing, 17 (4), 363–374. https://doi.org/10.1002/nvsm.1432
Daellenbach, K., Thirkell, P., & Zander, L. (2013). Examining the influence of the individual in arts sponsorship decisions. Journal of Nonprofit and Public Sector Marketing, 25 (1), 81–104. https://doi.org/10.1080/10495142.2013.759819
Dalakas, V. (2009). Consumer response to sponsorships of the performing arts. Journal of Promotion Management, 15 (1/2), 204–211. https://doi.org/10.1080/10496490902837650
Davies, H. T. O., Nutley, S. M., & Smith, P. C. (2000). What works? Evidence-based policy and practice in public services . Bristol: The Policy Press.
Dell’era, C. (2010). Art for business: Creating competitive advantage through cultural projects. Industry & Innovation, 17 (1), 71–89. https://doi.org/10.1080/13662710903573844
Dietz, J., Antonakis, J., Hoffrage, U., Krings, F., Marewski, J. & Zehnder, C. (2014). Teaching Evidence-based management with a focus on producing local evidence. Accessed April 10, 2015, from https://serval.unil.ch/resource/serval:BIB_9EC91D46ECD7.P001/REF
Dion, D., & Arnould, E. (2011). Retail luxury strategy: Assembling Charisma through art and magic. Journal of Retailing, 87 (4), 502–520. https://doi.org/10.1016/j.jretai.2011.09.001
Durand, J.-C., De Gouveia, M. A., & Berman, G. (1996). Business sponsorship and fiscal incentives for culture in Brazil: Analysis of a recent experience. The European Journal of Cultural Policy, 3 (1), 27–37. https://doi.org/10.1080/10286639609358032
Estes, Z., Brotto, L., & Busacca, B. (2018). The value of art in marketing: An emotion-based model of how artworks in ads improve products evaluations. Journal of Business Research, 85 , 396–405. https://doi.org/10.1016/j.jbusres.2017.10.017
File, K. M., & Prince, R. (1998). Cause related marketing and corporate philanthropy in the privately held Enterprise. Journal of Business Ethics, 17 (14), 1529–1539. https://www.jstor.org/stable/25073987
Finkel, R. (2010). Re-imaging arts festivals through a corporate lens: A case study of business sponsorship at the Henley festival. Managing Leisure, 15 (4), 237–250. https://doi.org/10.1080/13606719.2010.508664
Fishel, D. (2002). Australian philanthropy and the arts: How does it compare? International Journal of Arts Management, 4 (2), 9–15.
Frank, B., & Geppert, K. (2004). Are small recipients overlooked by sponsors? An empirical note. Journal of Cultural Economics, 28 (2), 143–156. https://doi.org/10.1023/B:JCEC.0000019574.70428.c7
French, S. (1991). The corporate art of helping the arts. Public Relations Quarterly, 36 (3), 25–27.
Goodman, J. S., Gary, M. S., & Wood, E. R. (2014). Bibliographic search training for evidence-based management education: A review of relevant literatures. Academy of Management Learning & Education, 13 (3), 322–353. https://doi.org/10.5465/ambpp.2014.159
Gorn, G. (1982). The effects of music in advertising choice behaviour: A classical conditioning approach. Journal of Marketing, 46 (1), 94–101. https://doi.org/10.1177/002224298204600109
Hagdtvedt, H., & Patrick, V. M. (2008a). Art infusion: The influence of visual art on the perception and evaluation of consumer products. Journal of Marketing Research, 45 (3), 379–389. https://doi.org/10.1509/jmkr.45.3.379
Hagdtvedt, H., & Patrick, V. M. (2008b). Art and the brand: The role of visual art in enhancing brand extendibility. Journal of Consumer Psychology, 18 (3), 212–222. https://doi.org/10.1016/j.jcps.2008.04.010
Herranz de la Casa, J. M., Manfredi, J. L., & Cabezuelo-Lorenzo, F. (2015). Latest trends and initiatives in corporate social responsibility: A communicational analysis of successful cases of arts and culture in Spain. Catalan Journal of Communication and Cultural Studies, 7 (2), 217–229. https://doi.org/10.1386/cjcs.7.2.217_1
Hetsroni, A., & Tukachinsky, R. H. (2005). The use of fine art in advertising: A survey of Creatives and content analysis of advertisements. Journal of Current Issues & Research in Advertising, 27 (1), 93–107. https://doi.org/10.1080/10641734.2005.10505176
Hewitt, J. (2000). Posters of distinction: Art, advertising and the London, Midland, and Scottish railways. The MIT Press, 16 (1), 16–35. https://doi.org/10.1162/074793600300159574
Hjorth, D., & Pelzer, P. (2007). The fate of phaeton: Baroque art for management sake? Organization, 14 (6), 869–886. https://doi.org/10.1177/1350508407085413
Hoeken, H., & Ruikes, L. (2005). Art for Art’s sake? An exploratory study of the possibility to align works of art with an Organization’s identity. International Journal of Business Communication, 42 (3), 233–246. https://doi.org/10.1177/0021943605277111
Huettl, V., & Gierl, H. (2012). Visual art in advertising: The effects of utilitarian vs. hedonic product positioning and price information. Marketing Letters, 23 (3), 893–904. https://doi.org/10.1007/s11002-012-9196-z
Hüttl-Maack, V. (2018). Visual art in advertising: New insights on the role of consumers’ art interest and its interplay with the hedonic value of the advertised product. Journal of Product & Brand Management, 27 (3), 262–276. https://doi.org/10.1108/JPBM-02-2017-1424
Jain, R., & Bagdare, S. (2011). Music and consumption experience: A review. International Journal of Retail and Distribution Management, 39 (4), 289–302. https://doi.org/10.1108/09590551111117554
Jeffri, J. (1989). Training the new Philantropists. Journal of Arts, Management and Law, 19 (2), 11–21. https://doi.org/10.1080/07335113.1989.9943117
Jelinek, J.-S. (2018). Art as strategic branding tool for luxury fashion brands. Journal of Product & Brand Management, 27 (3), 294–307. https://doi.org/10.1108/JPBM-01-2017-1408
Joy, A., Wang, J. J., Chan, T.-S., Sherry Jr., J. F., & Cui, G. (2014). M(art)worlds: Consumer perceptions of how luxury Brand Stores become art institutions. Journal of Retailing, 90 (3), 347–364. https://doi.org/10.1016/j.jretai.2014.01.002
Kapferer, J. N. (2014). The artification of luxury: From artisans to artists. Business Horizons, 57 (3), 371–380. https://doi.org/10.1016/j.bushor.2013.12.007
Kapferer, J.-N. (2012). Abundant rarity: The key to luxury growth. Business Horizons, 55 (5), 453–462. https://doi.org/10.1016/j.bushor.2012.04.002
Kawashima, N. (2012). Corporate support for the arts in Japan: Beyond emulation of the Western models. International Journal of Cultural Policy, 18 (3), 295–307. https://doi.org/10.1080/10286632.2011.651132
Kepes, S., Bennett, A. A., & Mcdaniel, M. A. (2014). Evidence-based management and the trustworthiness of our cumulative scientific knowledge: Implications for teaching, research, and practice. Academy of Management Learning & Education, 13 (3), 446–466. https://doi.org/10.5465/amle.2013.0193
Killacky, J. R. (1998). Corporate research and venture capital models for the arts. International Journal of Arts Management, 1 (1), 295–307. https://doi.org/10.1080/10286632.2011.651132
Kim, K., Ko, E., & Lee, Y. (2012). Art infusion in fashion product: The influence of visual art on product evaluation and purchase intention of consumers. Journal of Global Fashion Marketing, 3 (4), 180–186. https://doi.org/10.1080/20932685.2012.10600848
Kim, P., Vaidyanathan, R., Chang, H., & Stoel, L. (2018). Using brand alliances with artists to expand retail brand personality. Journal of Business Research, 85 , 424–433. https://doi.org/10.1016/j.jbusres.2017.10.020
Kirchberg, V. (1995). Arts sponsorship and the state of the City: The impact of local socio-economic conditions on corporate arts support. Journal of Cultural Economics, 19 (4), 305–320. http://www.jstor.org/stable/41810561
Kirchberg, V. (2004). Structures of corporate arts patronage between the world wars: A case study of the corporate leaders P.S. du Pont. Journal of Arts, Management and Law, 33 (4), 263–280. https://doi.org/10.3200/JAML.33.4.263-280
Koronaki, E., Kyrousi, A. G., & Panigyrakis, G. G. (2018). The emotional value of arts-based initiatives: Strengthening the luxury brand-consumer relationship. Journal of Business Research, 85 , 406–413. https://doi.org/10.1016/j.jbusres.2017.10.018
Kottasz, R., Bennett, R., Savani, S., Mousley, W., & Ali-Choudhury, R. (2007). The role of the corporate art collection in corporate identity management: The case of deutsche Bank. International Journal of Arts Management, 10 (1), 19–31. https://www.jstor.org/stable/41064905
Kottasz, R., Bennett, R., Savani, S., Mousley, W., & Ali-Choudhury, R. (2008). The role of corporate art in the management of corporate identity. Corporate Communications: An International Journal, 13 (3), 235–254. https://doi.org/10.1108/13563280810893634
Kressner Cobb, N. (1996). Looking ahead: Private sector giving to the arts and the humanities. The Journal of Arts Management, Law, and Society, 26 (2), 125–160. https://doi.org/10.1080/10632921.1996.9942959
Kressner Cobb, N. (2002). The new philanthropy: Its impact on funding arts and culture. Journal of Arts, Management and Law, 32 (2), 125–143. https://doi.org/10.1080/10632920209596969
Kristal, S., Baumgarth, C., & Henseler, J. (2018). “Brand play” versus “brand attack”: The subversion of brand meaning in non-collaborative co-creation by professional artists and consumer activists. Journal of Product & Brand Management, 27 (3), 334–347. https://doi.org/10.1108/JPBM-01-2017-1405
Lagier, J., & Godey, B. (2007). A scale for measuring aesthetic style in the field of luxury and art products. International Journal of Arts Management, 9 (2), 39–50. http://www.jstor.org/stable/41064918
Leclair, M. S., & Gordon, K. (2000). Corporate support for artistic and cultural activities: What determines the distribution of corporate giving? Journal of Cultural Economics, 24 (3), 225–241. https://doi.org/10.1023/A:1007686500896
Lee, J., Kim, J., & Yu, J. (2015a). Effects of congruence of product, visual image, and consumer self-image on art-infusion advertising. Social Behavior and Personality: An International Journal, 43 (10), 1725–1740. https://doi.org/10.2224/sbp.2015.43.10.1725
Lee, H. C., Chen, W. W., & Wang, C. W. (2015b). The role of visual art in enhancing perceived prestige of luxury brands. Marketing Letters, 26 (4), 593–606. https://doi.org/10.1007/s11002-014-9292-3
Lehu, J. (2011). Ceci n’est pas une marque. La Revue des Sciences de Gestion, 251 (5), 165–176. https://doi.org/10.3917/rsg.251.0165
Lewandowska, K. (2015). From sponsorship to Partnership in Arts and Business Relations. The Journal of Arts Management, Law and Society, 45 (1), 33–50. https://doi.org/10.1080/10632921.2014.964818
Lewandowska, K. (2016). It’s not all about the profit: An analysis of changes in arts and business relations. Economics and Business Review, 2 (16). https://doi.org/10.18559/ebr.2016.1.7
Lindenberg, M., & Oosterlinck, K. (2011). Art collections as a strategy tool: A typology based on the Belgian financial sector. International Journal of Arts Management, 13 (3), 4–19.
Martorella, R. (1996). Arts and business: an international perspective on sponsorship. Westport, CT: Greenwood Publishing Group.
Mcguigan, J., & Gilmore, A. (2002). The millennium dome: Sponsoring, meaning and visiting. The European Journal of Cultural Policy, 8 (1), 1–20. https://doi.org/10.1080/10286630290032486
McIsaac, P. (2007). Public-private support of the arts and German cultural policy the case of Wilhelm bode. International Journal of Cultural Policy, 13 (4), 371–391. https://doi.org/10.1080/10286630701683276
McNicholas, B. (2004). Arts, culture and business: A relationship transformation, a nascent field. International Journal of Arts Management, 7 (1), 57–68. http://www.jstor.org/stable/41064831
Megehee, C. M., & Spake, D. F. (2012). Consumer enactments of archetypes using luxury brands. Journal of Business Research, 65 (10), 1434–1442. https://doi.org/10.1016/j.jbusres.2011.10.009
Mermiri, T. (2010). Arts sponsorship: The facts, trends and potential. Journal of Sponsorship, 3 (4), 307–320.
Moir, L., & Taffler, R. J. (2004). Does corporate philanthropy exist? Business giving to the arts in the U.K. Journal of Business Ethics, 54 (2), 149–161. http://www.jstor.org/stable/25123333
Moon, J., & Kwak, J. (2010). The effect of art-parody and art-infusion advertisements focusing on product type and regulatory focus. Asian Journal of quality , 79–88. https://doi.org/10.1108/15982681011051840
Morbey, M. L. (2006). Killing a culture softly: Corporate partnership with a Russian museum. International Journal of Museum Management and Curatorship, 21 (4), 267–282. https://doi.org/10.1016/j.musmancur.2006.09.001
Morel, C. (2005). Will business ever become legitimate partners in the financing of the arts in France? International Journal of Cultural Policy, 11 (2), 199–213. https://doi.org/10.1080/10286630500198229
Mulcahy, K. V. (1999). Cultural patronage in the United States. International Journal of Arts Management, 2 (1), 53–58. https://www.jstor.org/stable/41064675
Naletelich, K., & Paswan, A. K. (2018). Art infusion in retailing: The effect of art genres. Journal of Business Research. https://doi.org/10.1016/J.JBUSRES.2017.10.030
Nam, J., & Shin, H. D. (2015). Corporate art support through foundations. Actual Problems of Economics, 172 (10), 304–315.
Nutley, S. (2003). Increasing research impact: Early reflections from the ESRC Evidence Network. ESRC UK Centre for Evidence Based Policy and Practice , Working Paper no.16. Accessed the 15 June, 2020 from https://www.kcl.ac.uk/sspp/departments/politicaleconomy/research/cep/pubs/papers/assets/wp16.pdf
Nutley, S., Davies, H. & Walter, I. (2002). Evidence Based Policy and Practice: Cross Sector Lessons From the UK. ESRC UK Centre for Evidence Based Policy and Practice , Working Paper no.9.
O’Hagan, J., & Harvey, D. (2000). Why do companies sponsor arts events? Some evidence and a proposed classification. Journal of Cultural Economics, 24 (3), 205–224. https://doi.org/10.1023/A:1007653328733
O’Reilly, D. (2011). Mapping the arts marketing literature. Arts Marketing: An International Journal, 1 (1), 26–38. https://doi.org/10.1108/20442081111129851
Olkkonen, R., & Tuominen, P. (2006). Understanding relationship fading in cultural sponsorship. Corporate Communications: An International Journal, 11 (1), 64–77. https://doi.org/10.1108/13563280610643561
Olkkonen, R., & Tuominen, P. (2008). Fading configurations in inter-organizational relationships: A case study in the context of cultural sponsorship. Journal of Business & Industrial Marketing, 23 (3), 203–212. https://doi.org/10.1108/08858620810858463
Peluso, A. M., Pino, G., Amatulli, C., & Guido, G. (2017). Luxury advertising and recognizable artworks. European Journal of Marketing, 51 (11/12), 2192–2206. https://doi.org/10.1108/EJM-09-2016-0496
Pelzer, P. (2006). Art for Management’s sake? A doubt. Culture & Organization, 12 (1), 65–77. https://doi.org/10.1080/14759550500490592
Pfeffer, J. & Sutton, B. (2015). Five principles of EBM . Accessed March 15, 2015, from http://evidence-basedmanagement.com/
Podsakoff, P. M., Mackenzie, S. B., Lee, J. Y., & Podsakoff, N. P. (2003). Common method biases in behavioral research: A critical review of the literature and recommended remedies. Journal of Applied Psychology, 88 (5), 879–903. https://doi.org/10.1037/0021-9010.88.5.879
Podsakoff, P. M., Mackenzie, S. B., Bachrach, D. G., & Podsakoff, N. P. (2008). The influence of management journals in the 1980s and 1990s. Strategic Management Journal, 26 , 473–488. https://doi.org/10.1002/smj.454
Polonsky, M., Garma, R., & Mittelstaedt, J. D. (2006). An examination of the globalization of authorship in publishing in 20 leading marketing journals. European Business Review, 18 (6), 437–456. https://doi.org/10.1108/09555340610711076
Preece, S. B. (2010). Building an arts-business partnership: The case of Aeroplan and tapestry new opera. International Journal of Arts Management, 12 (2), 49–58. https://doi.org/10.2307/41065018
Quester, P. G., & Thompson, B. (2001). Advertising and promotion leverage on arts sponsorship effectiveness. Journal of Advertising Research, 41 (1), 33–47. https://doi.org/10.2501/JAR-41-1-33-47
Reiss, A. (1989). Business takes a partner–the arts. Management Review, 78 (6).
Rentschler, R., & Kirchner, T. A. (2012). Arts management/marketing journal citation analysis: Assessing external impact. Arts Marketing: An International Journal, 2 (1), 6–20. https://doi.org/10.1108/20442081211232981
Rentschler, R., & Shilbury, D. (2008). Academic assessment of arts Management Journals: A multidimensional rating survey. International Journal of Arts Management, 10 (3), 60–71. https://www.jstor.org/stable/41064964
Riot, E., Chamaret, C., & Rigaud, E. (2013). Murakami on the bag: Louis Vuitton’s decommoditization strategy. International Journal of Retail & Distribution Management, 41 (11/12), 919–939. https://doi.org/10.1108/IJRDM-01-2013-0010
Roberts, N. C. & Bradley, R.T. (1999). Research methodology for new public management. International Public Management Network workshop in Siena, Italy, July 28-30, 1999. Accessed March 10, 2015, from http://www.rrojasdatabank.info/roberts.pdf
Robson, A. D. (2015). Industry: Art angel? Pepsi-Cola’s “portrait of America” art annual as an early instance of corporate art sponsorship. The Journal of American Culture, 38 (4), 329–343. https://doi.org/10.1111/jacc.12437
Roodhouse, S. (1997). Interculturism: The relationship between art and industry. Journal of Arts, Management and Law, 27 (3), 227–237. https://doi.org/10.1080/10632929709596965
Roschwalb, S. A. (1990). Corporate eyes on the market: Funding the arts for the 1990s. Journal of Arts, Management and Law, 19 (4), 73–84. https://doi.org/10.1080/07335113.1989.10593789
Ruth, J. A., & Simonin, B. L. (2003). Brought to you by brand a and brand B: Investigating multiple sponsors’ influence on consumers’ attitudes toward sponsored events. Journal of Advertising, 32 (3), 19–30. http://www.jstor.org/stable/4622165
Ryan, A., & Fahy, J. (2003). A relationship marketing perspective on the sponsorship of the arts in Ireland: A Galway arts festival-Nortel networks case study. Irish Marketing Review, 16 (1), 31–42.
Ryan, N. (2007). Prada and the art of patronage. Fashion Theory – Journal of Dress Body and Culture, 11 (1), 7–24. https://doi.org/10.2752/136270407779934588
Sauvanet, N. (1999). Sponsorship in France. International Journal of Arts Management, 21 (1), 59–63. https://www.jstor.org/stable/41064676
Scaltsa, M. (1992). Defending sponsorship and defending the responsibility of governments towards the visual arts. International Journal of Museum Management and Curatorship, 11 (4), 387–392. https://doi.org/10.1016/0964-7775(92)90077-I
Schroeder, J. E. (2005). The artist and the brand. European Journal of Marketing, 39 (11/12), 1291–1305. https://doi.org/10.1108/03090560510623262
Schwaiger, M., Sarstedt, M., & Taylor, C. R. (2010). Art for the sake of the corporation Audi, BMW group, DaimlerChrysler, Montblanc, Siemens, and Volkswagen help explore the effect of sponsorship on corporate reputations. Journal of Advertising Research, 50 (1), 77–90. https://doi.org/10.2501/S0021849910091208
Sciarelli, S. (2011). La gestione dell’impresa . Padova: Cedam.
Sherry Jr., J. F., & Schouten, J. W. (2002). A role for poetry in consumer research. Journal of Consumer Research, 29 , 218–234. https://doi.org/10.1086/341572
Stenstrom, E. (2008). What turn will cultural policy take? The renewal of the Swedish model. International Journal of Cultural Policy, 14 (1), 25–35. https://doi.org/10.1080/10286630701856476
Stern, B. B. (1991). Detailed image analysis: Poetic methodology for advertising research. International Journal of Advertising: the Review of Marketing Communications, 10 (2), 161–180. https://doi.org/10.1080/02650487.1991.11104446
Strauß, A. (2018). Value-creation processes in artistic interventions and beyond: Engaging conflicting orders of worth. Journal of Business Research, 85 , 540–545. https://doi.org/10.1016/j.jbusres.2017.10.033
Sylvestre, C. M., & Moutinho, L. (2007). Leveraging associations: The promotion of cultural sponsorships. Journal of Promotion Management, 13 (3/4), 281–303. https://doi.org/10.1080/10496490802308497
Tahai, A., & Meyer, M. J. (1999). A revealed preference study of management journals’ direct influences. Strategic Management Journal, 20 , 279–296. https://doi.org/10.1002/(SICI)1097-0266(199903)20:3<279::AID-SMJ33>3.0.CO;2-2
Thomas, S. R., Pervan, S. J., & Nuttall, P. J. (2009). Marketing orientation and arts organisations: The case for business sponsorship. Marketing Intelligence and Planning, 27 (6), 736–752. https://doi.org/10.1108/02634500910988654
Thompson, B. J. (2005). Sponsorship as a bilateral relationship: The benefits of applying relationship marketing principles in the sponsorship exchange. Asia Pacific Journal of Arts and Cultural Management, 3 (1), 188–203.
Tranfield, D., Denyer, D., & Smart, P. (2003). Towards a methodology for developing evidence-informed management knowledge by means of systematic review. British Journal of Management, 14 , 207–222. https://doi.org/10.1111/1467-8551.00375
Treccani. (2015). Arte . Accessed April 15, 2015, from http://www.treccani.it/vocabolario/arte/
Turgeon, N., & Colbert, F. (1992). The decision process involved in corporate sponsorship for the arts. Journal of Cultural Economics, 16 (1), 41–51. http://www.jstor.org/stable/41810470
Tweedy, C. (1991). Sponsorship of the arts-an outdated fashion or the model of the future? International Journal of Museum Management and Curatorship, 10 (2), 161–166.
Tyrie, A., & Ferguson, S. (2013). Understanding value from arts sponsorship: A social exchange theory perspective. Arts Marketing: An International Journal, 3 (2), 131–153. https://doi.org/10.1108/AM-10-2012-0018
Ulibarri, C. A. (2000). Rational philanthropy and cultural capital. Journal of Cultural Economics, 24 (2), 135–146. https://doi.org/10.1023/A:1007639601013
Van der Burg, T., Dolfsma, W., & Wilderom, C. P. M. (2004). Raising private investment funds for museums. International Journal of Arts Management, 6 (3).
Van Niekerk, A. And Conradie, M. (2016). Branding through art: The commercial value of visual and linguistic signs of art. Critical Arts, South-North Cultural and Media Studies, Vol. 30, Issue 2, pp. 233 – 251. doi: https://doi.org/10.1080/02560046.2016.1187795
Vanhaverbeke, W. (1992). How students evaluate business sponsorship of the arts in Flanders. Journal of Cultural Economics, 16 (1), 53–66.
Vukadin, A., Wongkitrungrueng, A., & Assarut, N. (2018). When art meets mall: Impact on shopper responses, Journal of product & Brand Management. Vol., 27 (3), 277–293. https://doi.org/10.1108/JPBM-01-2017-1406
Walliser, B. (2003). L’évolution et l’état de l’art de la recherche internationale sur le parrainage. Recherche et Applications en Marketing, 18 (1), 65–94. https://www.jstor.org/stable/40589356
Wang, Y. J., Cruthirds, K. W., Axinn, C. N., & Guo, C. (2013). In search of aesthetics in consumer marketing: An examination of aesthetic stimuli from the philosophy of art and the psychology of art. Academy of Marketing Studies Journal, 17 (2), 37–55.
Weinstein, L., & Cook, J. (2011). The benefits of collaboration between for-profit businesses and nonprofit arts- or culture- oriented organizations. SAM Advanced Management Journal, 76 (3), 4–9.
Wu, C.-T. (2003). Privatising culture: Corporate art intervention since the 1980s. London: Verso.
Wyszomirski, M. J. (1996). Where do we go from Here? The future of support for the arts and humanities. The Journal of Arts Management, Law, and Society, 26 (2). https://doi.org/10.1080/10632921.1996.9942955
Yoon, H., & Shin, H. D. (2014). Determinants of the number of artworks in corporate art collections. International Journal of Arts Management, 16 (2), 19–28.
Zolberg, V. (2000). Privatization: Threat or promise for the arts and humanities? International Journal of Cultural Policy, 7 (1). https://doi.org/10.1080/10286630009358130
Zorn, E., & Koidl, R. (1991). Exhibition marketing—The relationship between industry and the museums. Museum Management and Curatorship, 10 (2). https://doi.org/10.1016/0260-4779(91)90012-M
Zsolnai, L., & Wilson, D. (2018). Art-based business. Journal of Cleaner Production, 135 (1), 1534–1538.
Download references
Author information
Authors and affiliations.
IPAG Business School, Nice, France
Stefania Masè
You can also search for this author in PubMed Google Scholar
Rights and permissions
Reprints and permissions
Copyright information
© 2020 The Editor(s) (if applicable) and The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Masè, S. (2020). Systematic Literature Review in the Field of Art and Business. In: Art and Business. International Series in Advanced Management Studies. Springer, Cham. https://doi.org/10.1007/978-3-030-51769-4_3
Download citation
DOI : https://doi.org/10.1007/978-3-030-51769-4_3
Published : 30 August 2020
Publisher Name : Springer, Cham
Print ISBN : 978-3-030-51768-7
Online ISBN : 978-3-030-51769-4
eBook Packages : Business and Management Business and Management (R0)
Share this chapter
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Publish with us
Policies and ethics
- Find a journal
- Track your research

IMAGES
VIDEO
COMMENTS
Literature reviews establish the foundation of academic inquires. However, in the planning field, we lack rigorous systematic reviews. In this article, through a systematic search on the methodology of literature review, we categorize a typology of literature reviews, discuss steps in conducting a systematic literature review, and provide suggestions on how to enhance rigor in literature ...
Method details Overview. A Systematic Literature Review (SLR) is a research methodology to collect, identify, and critically analyze the available research studies (e.g., articles, conference proceedings, books, dissertations) through a systematic procedure [12].An SLR updates the reader with current literature about a subject [6].The goal is to review critical points of current knowledge on a ...
Systematic review vs. literature review. A literature review is a type of review that uses a less systematic and formal approach than a systematic review. Typically, an expert in a topic will qualitatively summarize and evaluate previous work, without using a formal, explicit method. ... Experts: Contact experts in the field to ask if they have ...
Systematic reviews: Structure, form and content. This article aims to provide an overview of the structure, form and content of systematic reviews. It focuses in particular on the literature searching component, and covers systematic database searching techniques, searching for grey literature and the importance of librarian involvement in the ...
Systematic literature reviews (SRs) are a way of synthesising scientific evidence to answer a particular research question in a way that is transparent and reproducible, while seeking to include all published evidence on the topic and appraising the quality of th is evidence. SRs have become a major methodology
A systematic review identifies and synthesizes all relevant studies that fit prespecified criteria to answer a research question (Lasserson et al. 2019; IOM 2011).What sets a systematic review apart from a narrative review is that it follows consistent, rigorous, and transparent methods established in a protocol in order to minimize bias and errors.
A preliminary review, which can often result in a full systematic review, to understand the available research literature, is usually time or scope limited. Complies evidence from multiple reviews and does not search for primary studies. 3. Identifying a topic and developing inclusion/exclusion criteria.
Guidance on Conducting a Systematic Literature Review Yu Xiao1 and Maria Watson1 Abstract Literature reviews establish the foundation of academic inquires. However, in the planning field, we lack rigorous systematic reviews. In this article, through a systematic search on the methodology of literature review, we categorize a typology of
A systematic literature review is a method which sets out a series of steps to methodically organize the review. In this paper, we present a guide designed for researchers and in particular early-stage researchers in the computer-science field. The contribution of the article is the following:•Clearly defined strategies to follow for a ...
The rationale for systematic literature reviews has been well established in some fields such as medicine for decades (e.g. Mulrow, 1994); however, there are still few methodological guidelines available in the management sciences on how to assemble and structure such reviews (for exceptions, see Denyer and Tranfield, 2009; Tranfield et al., 2003 and related publications).
Cite this article: Lame, G. (2019) 'Systematic Literature Reviews: An Introduction', in Pr oceedings of the 22nd International Conference on Engineering Design (ICED19), Delft, The Netherlands ...
A systematic review is a scholarly synthesis of the evidence on a clearly presented topic using critical methods to identify, define and assess research on the topic. A systematic review extracts and interprets data from published studies on the topic (in the scientific literature), then analyzes, describes, critically appraises and summarizes interpretations into a refined evidence-based ...
Systematic reviews have grown in numbers since they first emerged in the field of medicine in the late 1970s. 4, 6 Other types of scholarly reviews are often mistitled as a systematic review. 4 Conceptual overlap and muddiness exist among the many types of reviews found in the literature. 6 According to research by Martinic, 7 a complicating ...
The application of systematic or structured literature reviews (SLRs) has developed into an established approach in the management domain (Kraus et al. 2020), with 90% of management-related SLRs published within the last 10 years (Clark et al. 2021).Such reviews help to condense knowledge in the field and point to future research directions, thereby enabling theory development (Fink 2010 ...
Literature reviews can also be useful if the aim is to engage in theory development (Baumeister & Leary, 1997; Torraco, 2005). In these cases, a literature review provides the basis for building a new conceptual model or theory, and it can be valuable when aiming to map the development of a particular research field over time.
A literature review is a scholarly paper which provides an overview of current knowledge about a topic. It will typically include substantive findings, as well as theoretical and methodological contributions to a particular topic (Hart 2018, p. xiii).Traditionally in education 'reviewing the literature' and 'doing research' have been viewed as distinct activities.
Background Systematic literature searching is recognised as a critical component of the systematic review process. It involves a systematic search for studies and aims for a transparent report of study identification, leaving readers clear about what was done to identify studies, and how the findings of the review are situated in the relevant evidence. Information specialists and review teams ...
standalone literature review by providing a clear, detailed guide using the rigorous systematic literature review (SLR) methodology. In the information systems (IS) field, the past decade has seen a solid stream of research papers dedicated to helping improve the field's quality of literature reviews (Bandara et al., 2015; Levy & Ellis,
Abstract. Performing a literature review is a critical first step in research to understanding the state-of-the-art and identifying gaps and challenges in the field. A systematic literature review is a method which sets out a series of steps to methodically organize the review. In this paper, we present a guide designed for researchers and in ...
The so-called "review article" is a journal-length paper which has an overarching purpose to synthesize the literature in a field, without collecting or analyzing any primary data (Green, Johnson, ... Keele University & University of Durham; 2007. Guidelines for performing systematic literature reviews in software engineering. Kitsiou S ...
The systematic literature review is the most structured and methodical way of analysing academic contributions to a thematic field. However, the screening and analysis processes are both dependent on the individual decision making of the reviewers and it is always possible that valuable contributions are filtered out due to missing the ...
systematic literature review has three main focuses: tasks and sub-tasks (section3), evaluation metrics (section4), and ... Another field of evaluation concernshuman evaluation, which is deemed the gold standard to measure characteristics like fluency, faithfulness, and coherence in text generation systems [50, 59, 120, 122, 154]. ...
This systematic literature review targets two main audiences: early career researchers in natural language processing looking for an overview of the field and promising research directions, as well as experienced researchers seeking a detailed view of tasks, evaluation methodologies, open challenges, and recent mitigation strategies.
In this work, we present results from a systematic literature review of studies that supported creative learning using interactive environments with enactive characteristics, in educational contexts. ... For this purpose, we conducted a systematic literature review (SLR) in four academic bases in the field of Computer Science. This SLR has ...
Additionally, participants in the focus group suggest conducting field tests of the criteria in a municipal setting before scaling up to national policy frameworks. ... In this systematic literature review, a wide range of sustainable infrastructure topics were explored across the analysed articles. Three primary areas emerged: the role of ...
This systematic literature review qualitatively synthesized effectiveness and safety outcomes for treatments received in the real-world setting after 1 L CDK4/6i therapy in patients with HR+/ HER2- LABC/mBC. MEDLINE®, Embase, and Cochrane were searched using the Ovid® platform for real-world evidence studies published between 2015 and 2022. ...
In order to carry out a systematic literature review in the field of Art and Business, this work followed the steps indicated by the aforementioned authors, while nevertheless including a step of its own. A systematic literature review, which will be illustrated in detail, requires that the research be conducted in two phases: an initial ...
Method: A systematic literature review identified seven sustainability competencies. The assessment of ESD competency integration into the curriculum employed a two-tier scanning mechanism. In the initial step, subject outlines (SOs) were utilized to pinpoint sustainable subject learning outcomes (SLOs) and assessment learning outcomes (ALOs).
This study aimed to conduct an overview of systematic reviews in the field of violence against adults with disability. Eight electronic databases as well as gray literature from January 2022 to April 2023 were searched to identify systematic reviews that focused on violence against adults with disabilities.
Understanding the relationship between the intake of sugars and diet quality can inform public health recommendations. This systematic review synthesized recent literature on associations between sugar intake and diet quality in generally healthy populations aged 2 years or older. We searched databases from 2010 to 2022 for studies of any design examining associations between quantified sugar ...