Talk to our experts
1800-120-456-456
- CBSE Class 7 English Sample Paper-1 with Solutions (2018-19)
- Sample Papers


CBSE Sample Paper-1 for Class 7 English with Solutions - Free PDF Download
The Sample Paper of Class 7 English is easily available on the Vedantu website. This Class 7 English Sample Paper follows the pattern of CBSE markings and the syllabus included is solely based on the NCERT textbook for Class 7. In Class 7 as students are gearing to finish their middle school the CBSE pattern for exams also tests their knowledge in reading, writing, and grammar. And therefore students will be guided accordingly when they practice for their exams through the CBSE Sample Papers for Class 7 English. The link provided below is the solved Sample Paper of Class 7th English and is freely downloadable. Register Online for NCERT Solutions Class 7 Science tuition on Vedantu to score more marks in the CBSE board examination. Vedantu is a platform that provides free CBSE Solutions (NCERT) and other study materials for students. , s Students who are looking for better solutions can download Class 7 Math NCERT Solutions to help you to revise the complete syllabus and score more marks in your examinations.
Note: ➤ Calculate your potential NEET rank based on marks with our NEET Rank Predictor by Marks !

Free Download PDF Format - The Sample Paper For Class 7 English
Being ill-prepared for an exam is a student’s nightmare and one can avoid that by preparing for the exam using the link for the solved Class 7 English Sample Paper 2018-19. It is very easy for students to go through the Sample Paper with the important questions, reading comprehension, grammar, and writing skills. In the CBSE Sample Papers for Class 7 English, you will have a thorough understanding of all possible questions from both the ‘HoneyComb’ English textbook and the English Supplementary Reader ‘An Alien Hand’ for Class 7 English.
The Class 7 English Sample Paper link available above is very helpful as it can be printed and students can conveniently use it in hardcopy format. The downloadable link can be easily accessed even through other electronic devices. The students can choose according to their timing and benefit.
The Sample Paper For Class 7 English will help broaden your knowledge of English and that is very important for you not only for the annual exams but also for other fields throughout your life as well.
In the Sample Paper of Class 7 English, the questions from all chapters of the textbook ‘HoneyComb’. Like Three Questions, A Gift of Chappals, Gopal and the Hilsa Fish, The Ashes that Made Trees Bloom, Quality, Expert Detectives, The Invention of Vita – Wonk, Fire Friend, and Foe, A Bicycle in Good Repair, and The Story of Cricket.
The questions from the poems of the HoneyComb textbook are also a part of Class 7 English Sample Paper. And they are namely, The Squirrel, The Rebel, The Shed, Chivvy, Trees, Mystery of the Talking Fan, Dad and the Cat and the Tree, Meadow Surprises and Garden Snake.
In a sample of Class 7th, English at Vedantu platform students will also be provided with the questions from the chapters of the supplementary reader An Alien Hand. It includes all the chapters starting from The Tiny Teacher, Bringing Up Kari, The Desert, The Cop, and the Anthem, Golu Grows A Nose, I Want something In The Cage, Chandni, The Bear Story, A Tiger in The House, and the final chapter An Alien Hand.
Determiners, linking Words, adverbs (place and type), tense forms, clauses, passivation, adjectives (Comparative and Superlative), modal auxiliaries, word order in sentence types, and reported speech are the important part of the grammar syllabus for Class 7 English all included in the Sample Paper.
Once you have a fair knowledge of the English language you will know that it is indeed a very scoring subject.
When you develop your ability to read and understand the language you will find that it can be a confidence booster as well.
Some More Simple Yet Effective Tips to Improve Your English
Watching English movies is one of the best ways to improve vocabulary skills. This will help you to understand the language well and learn accurate pronunciation.
Read the newspaper and the magazines regularly. The new words can be noted down and learned the meanings as well. This will help you to use these words when you write an article or an essay in English. When you use new words the answers will be more impressive to the examiner to read.
Make a note of the new words and phrases that you come across. Try to have the conversation in English as much as possible.
Don't hesitate that you will go wrong. That is fine when you make mistakes you learn better.
Read the chapters thoroughly and understand. Sometimes you might not understand when you read it for the first time. Read it again and again. When you read you will improve and learn better.
Advantages of Vedantu
The explanations and the summary of the chapters are very easy to understand by all the students.
The chapters and the solutions are comprehensive.
Students can easily learn the accurate format of answering the questions.
The solutions are prepared by expert professionals having thorough subject knowledge.
They follow the latest CBSE curriculum so there is no possibility of any confusion.

FAQs on CBSE Class 7 English Sample Paper-1 with Solutions (2018-19)
1. How is the Sample Papers for Class 7 CBSE English Beneficial for me?
The CBSE Sample papers for class 7 English will give you accurate information about the marks weightage in the exams that will help you prepare for them accordingly. It will also help you in having a wide understanding of all the topics and the way the questions are formulated and appear in the exam. All in all, it will help you be ready for the upcoming exam and attain the top position in the class.
2. Which Section has the Highest Weightage in CBSE Class 7 English Subject?
The theory paper for class 7 CBSE English is divided into 3 sections the first one is for 20 marks that test the comprehension reading ability of the students and the questions are to be answered based on the given passage or a poem. The second section is for 30 marks which are to test the grammar knowledge and writing skills of the students. The final section is for 30 marks as well as to test the students textbook knowledge of students and their understanding of it.
3. What are the methods to Improve Writing Skills for the Class 7 CBSE English Exam?
Students should be clear with their literature and grammar. the language can be better only if it is grammatically correct. Practice reading and writing every day. When you do this you will improve on sentence formation and learn to write better
4. How can I make sure I am good at grammar?
Know all the rules of grammar. When you write something in English, get it checked by someone who is familiar with the grammatical rules of English. When writing, use active voice. Be clear about tenses, when to use present tense, past tense, and future tense. Use synonyms and antonyms to avoid the repetition of words. To be good at grammar you will also need practice, practice writing in English as much as you can to build a familiarity with the subject.
5. is Vedantu a reliable source?
Yes, Vedantu is a reliable source for the preparation of your exam. Vedantu provides a smooth learning process to all its students. You can get notes of various subjects on the website. Sample Papers and previous year’s papers are also provided to practice paper solving. Vedantu also provides free sessions to some of the students from time to time. As Vedantu is built with the help of phenomenal teachers who have an exceptional record of excellence you can take its help on all subject matters.
CBSE Class 7 Sample Papers
Cbse class 7 study materials.
7th Class Past papers
7th Class Past papers download PDF

Every institution in Pakistan is in charge of holding the 7th class annual term examination. The 7th class final examinations are held according to the school-issued schedule by the administration. Students looking for 7th class past papers are welcome to use this site. For your convenience, we have made the source of past papers available on our website, ilmkidunya.
You can visit the website any time and get the past papers there. When the date sheet is announced, the students resume their search for past papers. Past papers are an essential tool for you to finish your preparation. They contain every question from previous years' papers. With the help of past papers, you can get a pattern of papers before appearing in the finals.
Past Paper Usage
Past papers are an important tool for the preparation of exams. There are number of students who use past papers for their final studies. You can get a lot of hints and information from them. Students can get information regarding the paper pattern, timetable, and the choices include in the paper. The previous year's data is all contained in past publications. Past papers are available on our website for students who will appear in the 7th grade. Visit the website to verify it. You may easily start preparing for your finals using past papers. Additionally, you can get the 7th class Past Papers on the website in PDF format.
Benefits of having Past Papers.
For students at all educational levels, having access to past papers is really valuable. Past papers are prior papers that can be frozen for the upcoming classes to get an advantage on the paper type. Students can familiarize themselves with the previous question formats and expected levels of difficulty by going through these papers. By doing your preparation from the past papers, you can reduce your level of stress during the exam. You can solve each question by recalling them from the past paper that you studied. Past papers are a great way to learn how to revise effectively. Your revision can be improved from the past paper.
Additionally, students can practice and apply the knowledge and abilities they have learned during their studies by using past papers.
Frequently Asked Questions:
Q: 1 How to get the 7th class past papers online?
Ans: To get the 7th class past papers online, visit the website. Go to the section of past papers. There you have to select your class 7th to get your desired subject from past papers.
Q: 2 Do the past papers contain the information of time table?
Ans: Yes, the past papers contain information on the timetable. You can get the idea for your timings from them.
Q: 3 Are past papers a good source for study?
Ans: Yes, with the help of past papers, you can get an idea of paper pattern. You can also prepare for the exams in a better way by learning properly through past papers.

Is this page helpful?
Thank you for your feedback. We are happy our work helped you.
Now, please consider sharing this page to your friends/class-fellows. Thank you
We are sorry to know you could not find your required information.
Please take a moment to let us know about your feedback.
More on 7TH CLASS
Become 7TH CLASS Expert on ilmkidunya

If you are a teacher of Chemistry or BS Chemisty, you can join as a moderator of this BS Chemistry section on ilmkidunya.
The process is so simple: simply send you brief details/CV on whatsapp 0309-3809111 and our team will get back to you. Click here for more details .
FREE Advice on 7TH CLASS Admissions
.jpg)
to continue to ilmkidunya.com
Hi SoftSolutions
A vibrant community of 400,000 followers. Join to get daily updates for exams & study activities.
Join our 1.9 Million subscribers on Pakistan's #1 Educational Youtube Channel to keep learning.
A great way to remain in touch with latest educational news. Become one of our 60K+ followers.
Make your Insta feed full of interesting information & educational content. Follow us on Instagram.
Make your fun time more valuable. Get short informative & educational videos.
Copyright © 2024 ilmkidunya.com. No content of this website can be copied or reproduced in any form without permission.
Forgot Password
Register type.
Please Provide following information to Register

Essay Writing For Class 7 Format, Examples, Topics, Exercises
Essay writing is an essential skill that every student should develop as it helps to express thoughts and ideas in a structured and logical manner. Writing an essay can be a challenging task, especially for Class 7 students who are still in the process of learning and developing their writing skills. However, with the right guidance and practice, students can master the art of essay writing. In this essay, we will discuss the format, examples, topics, and exercises for Class 7 essay writing.
Also Read: Essay Writing Topics For Class 6
Format of an Essay:
Before we discuss the format of an essay, it is essential to understand what an essay is. An essay is a piece of writing that presents an argument or point of view on a specific topic. It is typically composed of an introduction, body paragraphs, and a conclusion.
The format of an essay varies depending on the type of essay and the teacher’s instructions. However, most essays follow a basic structure that includes the following:
1. Introduction: The introduction is the first paragraph of the essay that introduces the topic and provides some background information. It should also include a thesis statement that states the main argument or point of view of the essay.
2. Body Paragraphs: The body paragraphs are the middle section of the essay that presents the main arguments and evidence to support the thesis statement. Each paragraph should focus on a single point and include supporting details, examples, and evidence to back up the argument.
3. Conclusion: The conclusion is the final paragraph of the essay that summarizes the main points of the essay and restates the thesis statement in a new way. It should also provide a closing statement that leaves a lasting impression on the reader.
Examples of Essays:
Here are some examples of essays that Class 7 students can use as a reference:
1. My Favorite Animal:
Introduction: Introduce the topic of your favorite animal and why you chose it.
Body Paragraphs: Describe the physical characteristics of the animal, its behavior, and habits. Provide examples and evidence to support your arguments.
Conclusion: Summarize the main points of the essay and state why this animal is your favorite.
2. Advantages and Disadvantages of Technology:
Introduction: Introduce the topic of technology and provide some background information.
Body Paragraphs: Discuss the advantages and disadvantages of technology. Include specific examples and evidence to support your arguments.
Conclusion: Summarize the main points of the essay and state your opinion on whether the advantages outweigh the disadvantages or vice versa.
3. My Dream Vacation:
Introduction: Introduce the topic of your dream vacation and why you chose it.
Body Paragraphs: Describe the location, activities, and experiences you would like to have on your dream vacation. Include specific details and evidence to support your arguments.
Conclusion: Summarize the main points of the essay and state why this vacation is your dream.
Topics for Essays:
Here are some topics that Class 7 students can use to write essays:
1. My Favorite Season 2. My Best Friend 3. My Favorite Hobby 4. The Importance of Exercise 5. My Favorite Book 6. My Family 7. The Importance of Education 8. My Favorite Movie 9. The Importance of Recycling 10. My Favorite Sport
Essay Writing Exercises:
Here are some exercises that Class 7 students can use to practice their essay-writing skills:
1. Choose a topic and write a five-paragraph essay using the basic essay format. 2. Read a news article and write an essay summarizing the main points and providing your opinion on the topic. 3. Write an essay describing a historical event and its impact on society. 4. Write an essay discussing the benefits and drawbacks of social media. 5. Write an essay describing your favorite food and explaining why it is your favorite.
Essay writing is a vital skill that every Class 7 student should develop. By following the basic essay format and practicing with different topics and exercises, students can improve their writing skills and express their thoughts and ideas in a clear and concise manner.
It is also essential for students to read and analyze different types of essays to understand the structure, language, and tone used in different types of essays. With consistent practice and guidance from their teachers, students can become proficient writers and excel in their academic and personal lives.
HIGH SCHOOL
- ACT Tutoring
- SAT Tutoring
- PSAT Tutoring
- ASPIRE Tutoring
- SHSAT Tutoring
- STAAR Tutoring
GRADUATE SCHOOL
- MCAT Tutoring
- GRE Tutoring
- LSAT Tutoring
- GMAT Tutoring
- AIMS Tutoring
- HSPT Tutoring
- ISAT Tutoring
- SSAT Tutoring
Search 50+ Tests
Loading Page
math tutoring
- Elementary Math
- Pre-Calculus
- Trigonometry
science tutoring
Foreign languages.
- Mandarin Chinese
elementary tutoring
- Computer Science
Search 350+ Subjects
- Video Overview
- Tutor Selection Process
- Online Tutoring
- Mobile Tutoring
- Instant Tutoring
- How We Operate
- Our Guarantee
- Impact of Tutoring
- Reviews & Testimonials
- Media Coverage
- About Varsity Tutors
FREE Common Core: 7th Grade English Language Arts Practice Tests
All common core: 7th grade english language arts resources, free common core: 7th grade english language arts diagnostic tests, common core: 7th grade english language arts diagnostic test 1, free common core: 7th grade english language arts practice tests, practice tests by concept, language practice test, capitalization, punctuation, and spelling: ccss.ela-literacy.l.7.2, separate coordinate adjectives with a comma: ccss.ela-literacy.l.7.2.a, use correct spelling: ccss.ela-literacy.l.7.2.b, english grammar and usage in writing and speaking: ccss.ela-literacy.l.7.1, explain role of phrases and clauses in general and in sample sentences: ccss.ela-literacy.l.7.1.a, use phrases and clauses in sentences, correcting misplaced and dangling modifiers: ccss.ela-literacy.l.7.1.c, use simple, compound, complex, and compound-complex sentences: ccss.ela-literacy.l.7.1.b, meanings of unknown and multiple-meaning words and phrases: ccss.ela-literacy.l.7.4, consult print and digital reference materials to define and pronounce words: ccss.ela-literacy.l.7.4.c, use context clues to determine word meanings: ccss.ela-literacy.l.7.4.a, understand nuanced word meanings and relationships: ccss.ela-literacy.l.7.5, distinguish connotations among words with similar denotations: ccss.ela-literacy.l.7.5.c, interpret figures of speech in context: ccss.ela-literacy.l.7.5.a, reading practice test, craft and structure practice test, reading to determine author's point of view practice test, reading to understand structure practice test, reading to understand vocabulary in context practice test, integration of knowledge and ideas practice test, reading to compare and contrast texts practice test, reading to evaluate the argument and specific claims in a text practice test, key ideas and details practice test, reading to analyze elements of a text practice test, reading to cite textual evidence practice test, reading to determine main idea or theme practice test, practice quizzes, common core: 7th grade english language arts problem set 23, common core: 7th grade english language arts problem set 22, common core: 7th grade english language arts problem set 21, common core: 7th grade english language arts problem set 20, common core: 7th grade english language arts problem set 19, common core: 7th grade english language arts problem set 18, common core: 7th grade english language arts problem set 17, common core: 7th grade english language arts problem set 16, common core: 7th grade english language arts problem set 15, common core: 7th grade english language arts problem set 14, common core: 7th grade english language arts problem set 13, common core: 7th grade english language arts problem set 12, common core: 7th grade english language arts problem set 11, common core: 7th grade english language arts problem set 10, common core: 7th grade english language arts problem set 9, common core: 7th grade english language arts problem set 8, common core: 7th grade english language arts problem set 7, common core: 7th grade english language arts problem set 6, common core: 7th grade english language arts problem set 5, common core: 7th grade english language arts problem set 4, common core: 7th grade english language arts problem set 3, common core: 7th grade english language arts problem set 2, common core: 7th grade english language arts problem set 1.

Kickstart Your Grade 7 Learning Odyssey with Testpapers"
Dive into your Grade 7 academic odyssey with Testpapers, your prime source for Grade 7 Past Exam Papers. Discover an array of [Subject] Grade 7 Study Guides, Updated 2023 Exam Papers, and [Subject] Exam Preparation Material. Access our Free Grade 7 Exam Resources and leverage [Subject] Academic Success Resources. Participate in a Community-Driven Exam Preparation initiative with Interactive [Subject] Learning Resources. Uncover our Comprehensive Grade 7 Study Material and engage with our thriving [Subject] Exam Community. Testpapers is your companion in paving a successful academic path in Grade 7.
Largest library of grade 7 CAPS exam papers for South African students, available in both English & Afrikaans. Over 300+ exam papers & memos for 11 subjects. All our exam papers are 100% free to access.
Please note there are few Afrikaans papers available
New exam papers coming soon
For long periods of time, education has been important to the development of the different scientific, moral, and ethical fields of humanity. In addition, it has been recognized by some governments as a human right, but in some places around the world; education is not accessible for everyone. Many people believe that having an educational system without any cost would mean a better-educated society, whereas others argue that this would not be possible. To develop a better-educated society, governments should establish a totally free education system for the following three reasons. The first and most important reason why a totally free education scheme means a better-educated society is equal opportunities. Foster (1993) explains that…show more content… Free education allows everyone to study but with low intensity or levels of education. For example, governments with low incomes would not have enough money to employ professional teachers or provide students with all the technological equipment necessary in their studies; it is too expensive. In addition, with free education, the number of students will be impressive. It is important to emphasize that education is not the only responsibility that governments have. They also economically support other public institutions. According to OECD (2008), the major challenge for countries is to secure sufficient funding levels to enable tertiary education institutions to meet the growing expectations of society and respond to the growing demand by students. However, education without cost allows people the same rights and opportunities necessary to the development of an educated society with moral and ethical… As a result, of economic growth is also important to highlight that it reduces family
Grade 7 past exam papers
Grade 7 2022 past exam papers, gr 7 exam papers, gr 7 2023 exam papers, grade 7 2023 exam papers, grade 7 test papers, grade 7 exam papers and memos, grade 7 past exam papers and memos, accessing afrikaans papers.
If you're seeing this message, it means we're having trouble loading external resources on our website.
If you're behind a web filter, please make sure that the domains *.kastatic.org and *.kasandbox.org are unblocked.
To log in and use all the features of Khan Academy, please enable JavaScript in your browser.
New content!
Review articles, unit 1: proportional relationships, unit 2: rates and percentages, unit 3: integers: addition and subtraction, unit 4: rational numbers: addition and subtraction, unit 5: negative numbers: multiplication and division, unit 6: expressions, equations, & inequalities, unit 7: statistics and probability, unit 8: scale copies, unit 9: geometry.
Welcome, Login to your account.
Recover your password.
A password will be e-mailed to you.

2023 Grade 7 Exams
Download all Grade 7 examinations on the links provided below.
GRADE 7 EXAMINATIONS SET 1
Grade 7 examinations set 2, grade 7 examinations set 3, grae 7 examinations set 4, grade 7 examinations set 5, grade 7 examinations set 6.
You cannot print the contents of this website.
- LESSON NOTES
- LESSON PLANS
- 2021 SCHEMES
- POWERPOINT NOTES
- FORM 1 EXAMS
- FORM 2 EXAMS
- FORM 3 EXAMS
- FORM 4 EXAMS
- COMPREHENSIVE 1-4
- TOPICAL QUESTIONS
- K.C.S.E SYLLABUS
- REVISION MOCKS
- K.C.S.E REVISION
- 2018 K.C.S.E PAPERS
- 2017 K.C.S.E PAPERS
- 2016 K.C.S.E PAPERS
- 2015 K.C.S.E PAPERS
- 2014 K.C.S.E PAPERS
- 2013 K.C.S.E PAPERS
- 2012 K.C.S.E PAPERS
- 2011 K.C.S.E PAPERS
- 2010 K.C.S.E PAPERS
- 2008 K.C.S.E PAPERS
- 1996-2009 K.C.S.E PAPERS
- TOPICAL PAST PAPERS
- SECONDARY F1-4
- REVISION NOTES STD 4-8
- SCIENCE NOTES STD 4-8
- SOCIAL STUDIES NOTES STD 4-8
- COMPREHENSIVE NOTES STD 4-8
- SCIENCE POWERPOINT
- SOCIAL STUDIES POWERPOINT
- K.C.P.E TOPICAL REVISION
- BEST & WORST INSHAS
- 2018 K.C.P.E PAST PAPERS
- 2000-2017 K.C.P.E PAPERS
- SCHEMES STD 4-8
- GRADE 1 EXAMS
- GRADE 2 EXAMS
- GRADE 3 EXAMS
- GRADE 1 NOTES & CLASS READERS
- GRADE 2 NOTES & CLASS READERS
- GRADE 3 NOTES & CLASS READERS
- GRADE 1 SCHEMES OF WORK
- GRADE 2 SCHEMES OF WORK
- GRADE 3 SCHEMES OF WORK
- GRADE 1 CURRICULUM
- GRADE 2 CURRICULUM
- GRADE 3 CURRICULUM
- GRADE 1-3 SYLLABUS

Your Best Education, TSC & Latest News Website
Grade 7 Free Exams: Junior Secondary Termly Exams and Answers
GRADE 7 TERM 2 EXAMS
GRADE 7 AGRIC TERM 2 OPENER .pdf GRADE 7 TERM 2 PERFORMING ARTS SCHEMES.docx (3).pdf GRADE 7 BUS TERM 2 OPENER EXAM .pdf GRADE 7 COMP TERM 2 OPENER EXAM .pdf GRADE 7 INT SCI TERM 2 OPENER EXAM .pdf GRADE 7 ENG TERM 2 OPENER .pdf GRADE 7 FRE TERM 2 OPENER .pdf GRADE 7 TERM 2 PHYSICAL EDUCATION SCHEMES (3).pdf GRADE 7 CRE TERM 2 OPENER EXAM .pdf GRADE 7 TERM 2 PRETECHNICAL STUDIES SCHEMES.docx (3).pdf GRADE 7 VA TERM 2 OPENER EXAM.pdf GRADE 7 HE TERM 2 OPENER EXAM .pdf GRADE 7 HMSC TERM 2 OPENER EXAM .pdf GRADE 7 LS TERM 2 OPENER EXAM .pdf GRADE 7 MAT TERM 2 OPENER EXAM .pdf GRADE 7 PA TERM 2 OPENER EXAM .pdf GRADE 7 PE TERM 2 OPENER EXAM.pdf GRADE 7 KIS TERM 2 OPENER EXAM.pdf GRADE 7 SST TERM 2 OPENER EXAM.pdf GRADE 7 TERM 2 AGRICULTURE SCHEMES.docx (3).pdf GRADE 7 PRE TECH TERM 2 OPENER.pdf GRADE 7 TERM 2 BUSINES STUDIES SCHEMES.doc (3).pdf GRADE 7 TERM 2 CRE SCHEMES (2).pdf GRADE 7 TERM 2 CRE SCHEMES.pdf GRADE 7 TERM 2 ENGLISH SCHEMES.docx (3).pdf GRADE 7 TERM 2 HEALTH EDUCATION SCHEMES.docx (3).pdf GRADE 7 TERM 2 HOME SCIENCE SCHEMES (3).pdf GRADE 7 TERM 2 KISWAHILI SCHEMES (3).pdf GRADE 7 TERM 2 LIFE SKILLS SCHEMES.docx (3).pdf GRADE 7 TERM 2 MATHEMATICS SCHEMES (3).pdf
GRADE 7 TERM 1 EXAMS
G7 CRE SERIES 1&2 ENDTERM 1 EXAMS (2).pdf G7 AGRIC SERIES 1&2 ENDTERM 1 EXAMS (2).pdf G7 BUS SERIES 1&2 ENDTERM 1 EXAMS (2).pdf G7 ENG SERIES 1 &2 ENDTERM 1 EXAMS (2).pdf G7 HE SERIES 1 &2 ENDTERM 1 EXAMS (2).pdf G7 HSCI SERIES 1&2 ENDTERM 1 EXAMS (2).pdf G7 INT SCI SERIES 1&2 ENDTERM 1 EXAMS (2).pdf G7 KIS SERIES 1&2 ENDTERM 1 EXAMS (2).pdf G7 LS SERIES 1 &2 ENDTERM 1 EXAMS (2).pdf G7 MAT SERIES 1&2 ENDTERM 1 EXAMS (2).pdf G7 PA SERIES 1&2 ENDTERM 1 EXAMS (2).pdf G7 PE SERIES 1 &2 ENDTERM 1 EXAMS (2).pdf G7 SST SERIES 1&2 ENDTERM 1 EXAMS (2).pdf GRADE 7 ASSESSMENT BOOK.pdf GRADE 7 COMPUTER SCIENCE ENDTERM 1 EXAM.pdf GRADE 7 JSS ENDTERM 1 SET 1 EXAM.pdf GRADE 7 PRETECH ET1 EXAM.pdf GRADE 7 VISUAL ARTS ENDTERM 1 EXAM.pdf
Business Studies Grade 7 CBC Free Schemes of Work
CBC Grade 7 Social Studies Schemes of Work Free Editable Word, PDF Downloads
Grade 7 CBC Free Notes and Schemes of work pdf; Junior Secondary
JSS Grade 7 CBC Novel list (Kiswahili and English set books)
Health Education Grade 7 CBC Free Schemes of Work
Computer Science Grade 7 CBC Schemes of Work For Junior Secondary
Free Grade 7 CBC Curriculum Designs, Notes & Schemes of Work [Junior Secondary]
Grade 7 CBC Performing Arts Complete Schemes of Work Free
Official Grade 7 CBC Assessment Report For Junior Secondary Schools
Grade 7 Free CBC Exams
Mathematics Grade 7 CBC Schemes of Work For Junior Secondary
Junior Secondary Core Subjects (Mandatory Subjects) in grade 7,8 and 9
Grade 7 Free CBC Schemes of Work {Updated Version}
Social Studies Grade 7 CBC Schemes of Work For Junior Secondary

CBC-Upper-Primary-Grade-6-9-Curriculum-Designs-KICD
Related Posts
Home science form 4 schemes of work – term 1-3.
May 16, 2024
CBC Grade 6 Social Studies Schemes of Work Free Editable Word, PDF Downloads
Simplified form 2 history lesson notes, 112 comments.
I loved your blog post.Thanks Again. Fantastic.
I really liked your blog article.Thanks Again. Want more.
I really liked your blog.Really looking forward to read more. Want more.
I value the blog article. Awesome.
I really like and appreciate your blog post.Really thank you! Much obliged.
I really like and appreciate your blog.Much thanks again. Really Cool.
Looking forward to reading more. Great post.Really looking forward to read more. Cool.
Thanks a lot for the blog post.Thanks Again. Will read on…
I really enjoy the blog post. Great.
Looking forward to reading more. Great post.Really thank you! Great.
I am so grateful for your blog article. Will read on…
Leave a Reply Cancel reply
Your email address will not be published. Required fields are marked *
Save my name, email, and website in this browser for the next time I comment.
- CBSE sample papers
- CBSE Class 7 Sample Papers
- CBSE Class 7 Science Sample Paper
CBSE Sample Paper for Class 7 Science
As exams are around the corner, students are all set to appear for their CBSE Class 7 final exams. After finishing their revisions, now it’s time to practise some sample papers to understand the exam pattern well. We provide sets of CBSE sample papers prepared by our expert teachers as per the guidelines for Class 7 Science. These sample papers cover marks distribution, important Science questions, and important diagrams that appear in school exams.
CBSE Sample Question Paper for Class 7 Science
Students should brush up their knowledge by practising these sample papers. This will help students to get rid of exam fear, develop confidence and make them ready to face their Class 7 final exams. Sample Papers provide a fair idea about the time taken to solve the actual CBSE question paper for class 7. With this, students can calculate their speed and devote time to each question in a planned manner. It will improve their time-management skills for the exam.
Here, we are providing the CBSE Class 7 science question papers without solutions which will give a fair idea of the real test paper and marking strategy set by the CBSE board. These question papers adhere to the CBSE Class 7 Syllabus and give you an in-depth understanding of each topic in a proper way. Students are advised to practise the class 7 science CBSE question papers thoroughly.
Students can look at the Summative assessment question paper of CBSE Class 7 Science below in the table. We have provided the SA1 and SA2 Science sample papers for students’ practice. These CBSE Sample Papers will also boost their exam preparation and help them score more marks in the exam.
Stay tuned for the latest updates on CBSE and other competitive exams. To get more sample papers, visit the CBSE Class 7 Sample Papers and get papers on other subjects. Download BYJU’S App and subscribe to the YouTube channel to access interactive study videos.
Leave a Comment Cancel reply
Your Mobile number and Email id will not be published. Required fields are marked *
Request OTP on Voice Call
Post My Comment
- Share Share
Register with BYJU'S & Download Free PDFs
Register with byju's & watch live videos.


30,000+ students realised their study abroad dream with us. Take the first step today
Meet top uk universities from the comfort of your home, here’s your new year gift, one app for all your, study abroad needs, start your journey, track your progress, grow with the community and so much more.

Verification Code
An OTP has been sent to your registered mobile no. Please verify

Thanks for your comment !
Our team will review it before it's shown to our readers.

- School Education /
7 Interesting and Fun English Stories For Class 3

- Updated on
- May 15, 2024

Stories that are interesting and fun make learning great for kids! Along with enjoying themselves and having fun, they also learn. English stories are a great way to ensure the holistic development of children. They also pick up skills like recitation, pronunciation, confidence, etc. Hence, in this blog, we bring you 7 Interesting and Fun English Stories For Class 3. Here, you will find some great stories for kids. Let’s explore them now!
Table of Contents
- 1 1. The Bear and the Two Friends
- 2 2. The Miser and His Gold
- 3 3. The Dog At the Well
- 4 4. Controlling Anger
- 5 5. The Leap at Rhodes
- 6 6. The Wolf and the Sheep
- 7 7. The Tortoise And The Bird
- 8 FAQs
1. The Bear and the Two Friends
Also Read : Mindfulness for Kids: With 11 Exercises, Importance
2. The Miser and His Gold
Also Read : 5 Tips to Improve Kid’s Vocabulary
3. The Dog At the Well
Moral : Heed the warnings of those who are wiser.
4. Controlling Anger
Also Read : 5 Best Fine Motor Activities for Preschoolers
5. The Leap at Rhodes
6. the wolf and the sheep , 7. the tortoise and the bird .
Ans: Some of the best stories are mentioned below: – The Golden Egg – The Shepherd Boy and the Wolf – Having a Best Friend: Friendship Moral Stories in English – The King’s Painting – The Pig and the Sheep
Ans: “Cinderella” is one of the most famous stories for kids.
Ans: A short story is simply written material consisting of a plot that may be fictional and can be read in a short time span of, like, 5-10 minutes.
Related Reads :
Hope you like the English stories for class 3 that we have provided in this blog. For more such kids’ learning material, check out School Education and follow Leverage Edu!!
Deepansh Gautam
Leave a Reply Cancel reply
Save my name, email, and website in this browser for the next time I comment.
Contact no. *


Connect With Us

30,000+ students realised their study abroad dream with us. Take the first step today.

Resend OTP in

Need help with?
Study abroad.
UK, Canada, US & More
IELTS, GRE, GMAT & More
Scholarship, Loans & Forex
Country Preference
New Zealand
Which English test are you planning to take?
Which academic test are you planning to take.
Not Sure yet
When are you planning to take the exam?
Already booked my exam slot
Within 2 Months
Want to learn about the test
Which Degree do you wish to pursue?
When do you want to start studying abroad.
January 2024
September 2024
What is your budget to study abroad?

How would you describe this article ?
Please rate this article
We would like to hear more.
Have something on your mind?

Make your study abroad dream a reality in January 2022 with
India's Biggest Virtual University Fair

Essex Direct Admission Day
Why attend .

Don't Miss Out

Class 12 Chemistry Answer Key 2024 Out, For Set 1, 2, 3
Students can match their answers with the most accurate CBSE Class 12 Chemistry Answer Key 2024 prepared by the expert facilities of Adda247. Get CBSE Chemistry Class 12 Question Paper Set 1, 2, 3.
Table of Contents
Chemistry Answer key 2024
The Central Board of Secondary Education has successfully conducted the CBSE Class 12 Chemistry Exam 2024 o n 27 February 2024. The chemistry paper was concluded at 01:30 pm, As the completion of the examination we updated the CBSE Class 12 Chemistry Answer Key 2024 on this page. The students appearing in the examination can match their answers with the unofficial Class 12 Chemistry Answer Key 2024 prepared by the expert facilities of Adda247.
Class 12 Chemistry Answer Key 2024
Here we have given the complete CBSE Class 12 Chemistry Answer Key 2024 along with Exam Analysis. In CBSE Class 12 Chemistry Answer Key & Exam Analysis, we will cover the difficulty level of the exam, out-of-syllabus questions, and mistakes in the question paper. CBSE Class 12 Chemistry Answer key 2024 is provided for all sections. Section A consists of 16 multiple-choice questions worth one mark each. Section B consists of five short answer questions worth two marks each. After completion of the CBSE Exam for the Chemistry subject, the students must match their responses with the Chemistry Answer Key provided on this page and must check the CBSE Class 12 Chemistry Exam Analysis.
Start your Preparation with CUET 2024 Arjun 2.0 Science Complete Batch
Answer Key Chemistry Class 12: Overview
CBSE Class 12 Chemistry Answer Key 2024 given on this page is error-free and all the answers are framed by the faculties who have years of experience in teaching Class 12 Chemistry. Below we have given the highlights of CBSE Answer Key Chemistry Class 12. The students must check the brief information of CBSE Class 12 Chemistry Answer Key 2024 provided in the table below.

CBSE Class 12 Chemistry Answer Key Set 1 (56/2/1)
SECTION A Questions no. I to 16 are Multiple Choice type Questions, carrying 1 mark each 16×1=16
1. When MnO2 is fused with KOH in air, it gives (A) KMnO4 (B) K2MnO4 (C) MngO7 (D) Mn2O Answer: (B) K2MnO4
2. Ligand EDTA is an example of a: (A) Monodentate ligand (B) Didentate ligand (C) Tridentate ligand (D) Polydentate ligand
Answer:(D) Polydentate ligand
3. Which of the following ligand forms chelate complex? (A) (CO2-) 4 (B) CI (C) NO (D) NH3
Answer:(A) CO 4

Answer: Option C

Answer: Option (d)
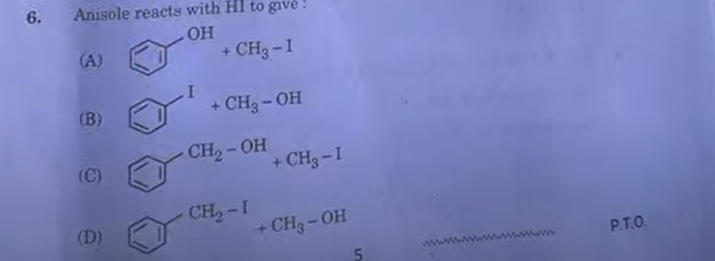
Answer: Option A
7. Ethanol on heating with conc, H₂SO₄ at 413 K gives: (A) C2H5OSO3H (B) C₂H-O-CH3 (C) C₂H-O-C2H5 (D) CH2=CH2 Answer:(C) C₂H-O-C2H5
8. An azeotropic solution of two liquids has boiling point lower than either of them when it : (A) is saturated (B) shows positive deviation from Raoult’s law (C) shows negative deviation from Raoult’s law (D) shows no deviation from Raoult’s law Answer:(B) shows positive deviation from Raoult’s law
9. The relative lowering of vapour pressure of an aqueous solution containing non-volatile solute is 0-0225. The mole fraction of the non-volatile solute is: (A) 0.80 (B) 0.725 (C) 0.15 (D) 0.0225 Answer: (D) 0.0225
10. During electrolysis of aqueous solution of NaCl: (A) H₂ (g) is liberated at cathode (B) Na is formed at cathode (C) O2 (g) is liberated at anode (D) Cl2 (g) is liberated at cathode
11. The addition of catalyst during a chemical reaction alters which of the following quantities of the reaction? (A) Enthalpy (B) Activation energy (C) Entropy (D) Internal energy
Answer: (B) Activation energy
12. For the elementary reaction PQ. the rate of disappearance of ‘P’ increases by a factor of 8 upon doubling the concentration of ‘P’. The order of the reaction with respect to ‘P’ is: (A) 3 (B) 4 (C) 2 (D) 1
Answer: (A) 3
For Questions number 13 to 16, two statements are given one labelled as Assertion (A) and the other labelled as Reason (R). Select the correct answer to these questions from the codes (A), (B), (C) and (D) as given below. (A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A). (B) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A). (C) Assertion (A) is true, but Reason (R) is false. (D) Assertion (A) is false, but Reason (R) is true.
13. Assertion (A): Aliphatic primary amines can be prepared by Gabriel phthalimide synthesis. Reason (R): Alkyl halides undergo nucleophilic substitution with anion formed by phthalimide.
Answer:(A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A). 14. Assertion (A): Uracil base is present in DNA. Reason (R): DNA undergoes self-replication.
Answer: (D) Assertion (A) is false, but Reason (R) is true. 15. Assertion (A): Diazonium salts of aromatic amines are more stable than those of aliphatic amines. Reason (R): Diazonium salts of aliphatic amines show resonance.
Answer:(A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A). 16. Assertion (A): p-nitroaniline is a weaker base than p-toluidine. Reason (R): The electron withdrawing effect of NO2 group in p-nitroaniline makes it a weaker base.
Answer:(A) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A).

Chemistry Set 2 Answer Key 2024: Paper Code (56/4/2)
3. Which alkyl halide Dom the given options will undergo a reaction

4. Which of the following stids represents Vitamin C? (A) Banelusric acid (B) Gluconic acid (C) Ascorbic acid (D) Benzoic acid

6. Consider the following reaction
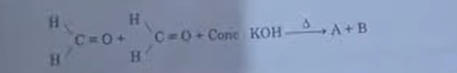
Identify A and B from the given options (A) A-Methanol. B-Potassium formate (B) A-Ethanol. B-Potassium formate (C) A-Methanal, 8-Ethanol (D) A-Methanol, B-Potassium acetate
Answer: (A) A-Methanol. B-Potassium formate
11. Nucleophilic addition of Grignard reagent to ketones followed by hydrolysis with dilute acids forms: (A) Alkene (C) Tertiary alcohol (B) Primary alcohol (D) Secondary alcohol
Answer: (C) Tertiary alcohol
12. Match the reagents required for the given reactions:
(A) 1-(г), П-(p), III-(s), IV(q) (B) 1-(q), II(r), III-(p). IV- (9) (C) 1-(8), II-(q). III-(p). IV(r) (D) 1-(p). II-(s), III-(r). IV- (q)
Answer: (A) 1-(г), П-(p), III-(s), IV(q)

Chemistry Set 3 Answer Key 2024: Paper Code (56/3/3-13)
1. Which of the following ligands is an ambidentate ligand? (A) CO (B) SCN- (C) NH3 (D) H₂O
Answer: (B) SCN-
2. On adding AgNO3 solution to 1 mole of CoCl3. 4NH3, one mole of AgCl is precipitated. The secondary valency of Cois: (A) 6 (C) 3 (B) 4 (D) 7
Answer: (A) 6
3. Which of the following elements of 3d series of transition elements has the lowest Δ a H°? (B) Cr (A) Sc (D) Zn (C) Cu
Answer: (D) Zn
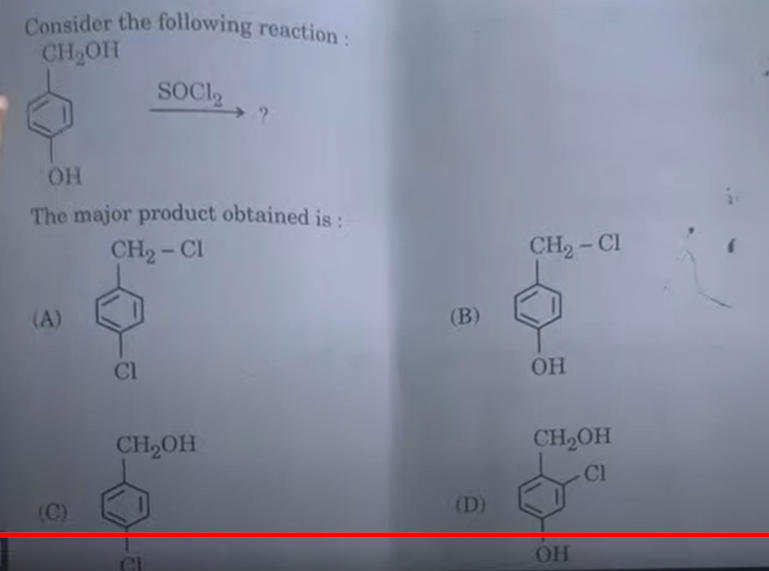
Answer: Option B

6. CH3-O-CH3 when treated with excess HI gives: (A) CH3-OH + CH3-1 (B) 2CH3-OH (C) 2CH3-I (D) CH3-I+CH4
Answer:C) 2CH3-I
Which of the following compounds will not react with benzene sulphonyl chloride? (A) (C2H5)3N (B) C2H5-NH2 (C) (C2H5)2 NΗ (D)C6H5- NH2
Answer:(A) (C2H5)3N
6. Scurvy’ is caused by a deficiency of vitamins: (A) E (B) A (C) C (D) D Answer: (C) C
7. A 1% solution of solute ‘X’ is isotonic with a 6% solution of sucrose (molar mass = 342 g mol¹). The molar mass of solute ‘X’ is: (A) 34-2 g mol-1 (B) 57 g mol-1 (C) 114 g mol-¹ (D) 3-42 g mol 1
10. The half life of a first order reaction with rate constant (k) of 3 min-1 (A) 0.693 min (B) 2.31 min (C) 6.93 min (D) 0.231 min
Answer: (D) 0.231 min
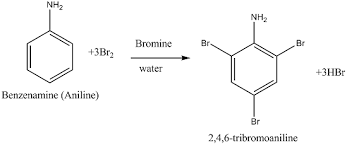
11. Which of the following cells is used in hearing aids? (A) Dry cell (B) Mercury cell (C) Nickel-cadmium cell (D) Fuel cell Answer: (C) Nickel-cadmium cell 12. Aniline on reaction with Bromine water gives: (A) o-bromoaniline (B) 2,4,6-tribromoaniline (C) m-bromoaniline (D) p-bromoaniline
Answer: (B) 2,4,6-tribromoaniline
13. Assertion (A): AmixH and AmixV are zero for an ideal solution. Reason (R): The solution which obeys Raoult’s law over the entire range of concentration is called an ideal solution. Answer: (a)
14. Assertion (A): Rate of reaction decreases with increase in temperature. Reason (R): Number of effective collisions increases with increase in temperature.
Assertion (A): Phenol on reaction with aqueous NaOH gives sodium phenoxide. Reason (R): This reaction supports the acidic nature of phenol.
Asertion (A): Boiling point of butan-1-ol is higher than that of butan-1-amine. Reason (R): Being more polar, butan-1-ol intermolecular hydrogen bonds form stronger compared to as butan-1-amine.
CBSE Class 12 Chemistry Answer Key 2024 Set 1
SECTION – A (MCQs)
1. Van’t Hoff factor for K₂SO4 solution, assuming complete ionization is
(A) 1 (B) 3 (C) 13 (D) 2
Answer: (B) 3
5. The rate of a reaction increases sixteen times when the concentration of the reactant increases four times. The order of the reaction is
(A) 2.5 (B) 2.0 (C) 1.5 (D) 0.5
Answer: (B) 2.0
6. Which of the following cell is used in hearing aids?
(A) Mercury cell (B) H2-O2 fuel cell (C) Dry cell (D) Ni-Cd cell
Answer: (A) Mercury cell
7. Isotonic solutions have the same
(A) density (C) osmotic pressure (B) refractive index (D) volume
Answer: (C) osmotic pressure
8. Transition metals are known to make interstitial compounds. Formation of interstitial compounds makes the transition metal
(A) more hard (C) more ductile (B) more soft (D) more metallic
Answer: (A) more hard
9. Auto-oxidation of chloroform in air and light produces a poisonous gas known as
(A) Phosphine (C) Phosgene (B) Mustard gas (D) Tear gas
Answer: (C) Phosgene
10. Out of the following alkenes, the one which will produce tertiary butyl alcohol on acid catalysed hydration is
(A) CH3CH3CH=CH2 (B) CH3CH=CH2 (C) CH3-CH=CH-CH3 (D) (CH3)2C = CH2
Answer: (D) (CH3)2C = CH2
CBSE Class 12 Chemistry Answer key 2024 Set 3
Check out the solutions of paper code 56/5/3 section A Mcqs in the table below.
CBSE Class 12th Chemistry Answer key 2024 Set 1
Check out the solutions of paper code 56/1/1 section A Mcqs in the table below.
Class 12 Chemistry Answer Key 2024 & Paper Analysis
Watch the video to get the Class 12 Chemistry Exam Analysis at the earliest.
Answer Key of Chemistry Class 12 CBSE 2024 for Set 1,2,3
Here we will provide the CBSE Class 12 Chemistry Answer key 2024 for Chemistry question sets 1, 2 and 3. A majority of students said that Section A’s Multiple Choice questions are a little bit tricky and they found some difficulties while solving these sections. Candidates can cross-check their answers with the answer key of chemistry class 12 cbse 2024 of Section A.
CBSE Class 12 Chemistry Board Question Paper 2024 PDF
The CBSE Class 12 Chemistry Exam was conducted in multiple sets at different exam centers. Students should assess the difficulty level of the Chemistry Question paper sets that differ from their own. The board question paper has a weightage of 70 points and all questions are mandatory to attempt. Students can access CBSE 12 Chemistry question papers from various sets on this page.
Class 12 Chemistry Question Paper Analysis 2024
After the conclusion of the Exam, we talked to the students as well as the teachers to know the difficulty level of the Paper. Let’s check the test takers’ and subject experts’ reactions on the CBSE Class 12 Chemistry Exam below.
CBSE Class 12 Chemistry Paper Analysis by Students
According to general reviews, students regarded the work to be moderate and balanced.
- The paper was NCERT-based and covered all relevant topics.
- The MCQ component of the paper was a little challenging but workable. Section B was easy.
- The long-answer questions were straightforward, if fairly lengthy.
- The short-answer questions were moderately difficult.
- Organic chemistry is frequently the most difficult component of the exam. Students rated organic chemistry this year as moderate. Expected questions regarding conversions and name reactions were posed.
CBSE Class 12th Chemistry Exam Analysis by Teachers
Experts and educators have stated that the chemistry paper was balanced and of moderate difficulty.
- The majority of the questions were direct and based on NCERT. Almost all of the students found the CBSE Class 12 Chemistry paper to be fairly average.
- The paper pattern was consistent with the CBSE sample paper.
- The majority of the paper’s questions ranged from simple to moderate, except for a couple that could have been difficult for pupils.
Class 12 Chemistry Question Paper 2024 Pattern
With calculating the scores with the CBSE Class 12 Chemistry Answer Key 2024 , students must know the Chemistry marking Scheme of Each section. As per the CBSE Class 12 Chemistry sample paper, the CBSE Class 12 Chemistry question paper consists of Five sections. All the sections hold different marks and different types of questions. The board will ask the very short answer type, short answer type, and case-based questions in the CBSE Class 12 Chemistry Examination. Check out the detailed section-wise pattern listed below:
1. Section A consists of 18 Questions carrying 1 mark each [Multiple Choice Questions]
2. Section B consists of 7 questions carrying 2 marks each [Very Short Answer Questions]
3. Section C consists of 5 questions carrying 3 marks each [Short Answer Questions]
4. Section D consists of 2 Questions carrying 4 marks each [Case Based Questons]
5. Section E consists of 2 Questions carrying 5 marks each [Long Answer Questions]
CBSE Class 12 Chemistry Answer Key Set 3 Last year
1, A compound CaCl2.6h2O undergoes complete dissociation in water, The Van’t Hoff Factor ‘i’ is:
a) 9, b) 6, c)3, d) . 4
Answer: c) 3
2. For a Zero order reaction of type A →products, the rate equation may be expressed as:

Answer: Option (a)
3. Which of the following Cu 2+ halide is not known?
Answer: Option (b)
Q4. Which of the following structures represents a-D-glucose?
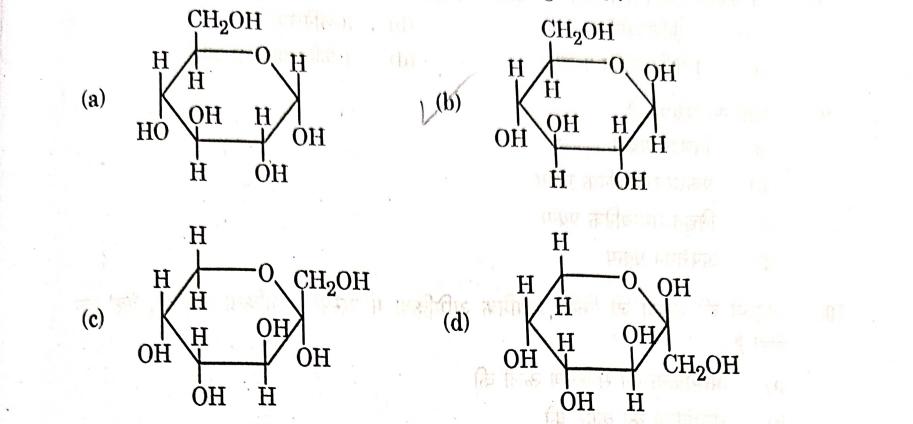
Answer: (a)
- The compounds [Cr(H2O)lCl3, [Cr(H2O) Cl]C12. H2O and [Cr(H2O)4C12]Cl. 2H2O exhibit:
(a) Linkage isomerism (b) Geometrical isomerism (c) . Ionization isomerism (d) Hydrate isomerism
Answer: (d) Hydrate isomerism
- Which of the following alkenes on acid catalysed hydration gives a tertiary alcohol?
(a) 2-Butene
(b) 2-Methylpropene
(c) Propene
(d) 95 1-Butene
Answer: (b) 2-Methylpropene
- When nitrobenzene is heated with tin and concentrated HCl, the product formed is:
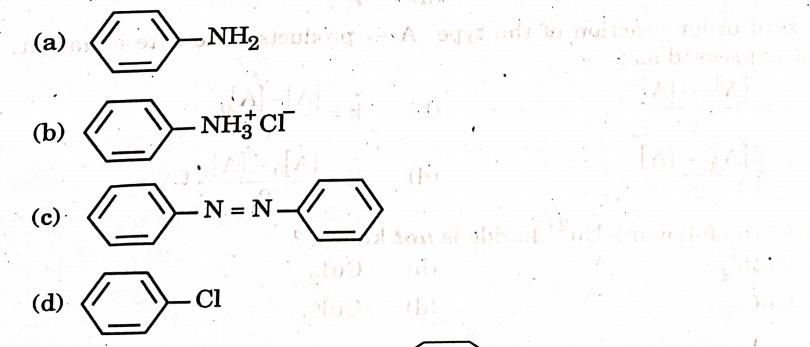
- The reaction of 1-phenyl-2-chloropropane with alcoholic KOH gives mainly:
(a) 1-phenylpropene
(b) 3-phenylpropene
(c) 1-phenylpropene 1-phenylpropan-3-ol
(d) 1-phenylypropan-2-ol
Answer: (a) 1-phenylpropene
- Corrosion of iron is:
(a) a decomposition process a photochemical process (b) a photochemical process (c) an electrochemical process (d) a reduction process
Answer: (a) reduction process
- The number of molecules that react with each other in an elementary reaction is a measure of the:
(a) activation energy of the reaction (b) order of the reaction (c) stoichiometry of the reaction (d) molecularity of the reaction
Answer: (d) molecularity of the reaction
11. On hydrolysis, which of the following carbohydrates gives glucose and galactose?
(a) Sucrose (c) Maltose (b) Lactose (d) Cellulose
Answer: (b) Lactose
12. The deficiency of which of the following vitamins causes ‘Rickets’?
(a) Vitamin A (b) Vitamin D (c) Vitamin B (d) Vitamin C
Answer; (b) Vitamin D
13. Which of the following is an ‘Acetal’?
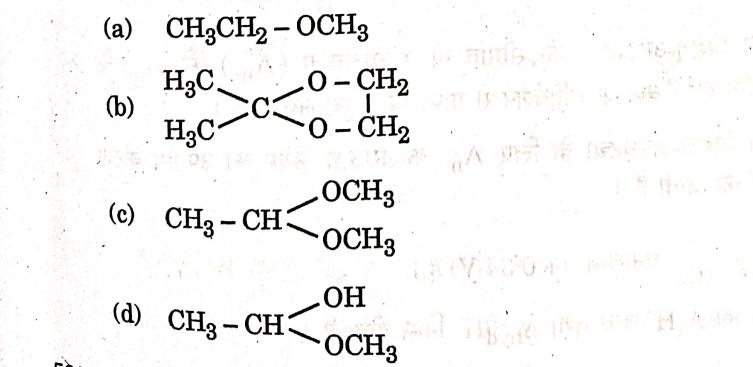
Answer: (d)
For Questions number 15 to 18, two statements are given – one labelled as Assertion (A) and the other labelled as Reason (R). Select the correct answer to these questions from the codes (a), (b), (c) and (d) as given below.
(a) Both Assertion (A) and Reason (R) are true and Reason (R) is the correct explanation of the Assertion (A). (b) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A). (c) Assertion (A) is true, but Reason (R) is false. (d) Assertion (A) is false, but Reason (R) is true.
- Assertion (A): When NaCl is added to water, a depression in freezing point is observed.
Reason (R): The vapour pressure of solution is increased which causes depression in freezing point.
Answer:(c) Assertion (A) is true, but Reason (R) is false.
- Assertion (A): Monobromination of aniline can be conveniently done by protecting the amino group by acetylation.
Reason (R): Acetylation decreases the activating effect of the amino group.

Class 12 Chemistry Answer Key last Year
- The magnetic moment of [NiCl4]2-
(a) 1.82 BM (b) 2.82 BM (c) 4.42 BM (d) 5.46 BM (Atomic no : Ni= 28) Answer: (a) 1.82 BM
- Which of the following cell was used in Apollo space programme?
(a) Mercury Cell
(b) Daniel Cell
(c) H2-O2 Fuel cell
(d) Dry Cell
Answer: (c) H2-O2 Fuel cell
- Which one of the following has lowest pKa value?
(a) CH3-COOH
(b) O2N-CH2-COOH
(c) Cl-CH2-COOH
Answer: (b) O2N-CH2-COOH
- Which of the following ions has the electronic configuration 3d6 ?
(a) Ni 3+ (b) Co 3+ (c) Mn 2+ (d) Mn 3+ Answer: (b) CO 3+
- Consider the following standard electrode potential values:
Fe3+ (aq) + e- —> Fe2+ (aq) Eo = + 0.77 V MnO4- (aq) + 8H+ + 5e- —-> Mn2+ (aq) + 4H2O (l) Eo = + 1.51V What is the cell potential for the redox reaction? (a) -2.28 V (b) – 0.74 V (c) + 0.74 V (d) + 2.28 V Answer: (c) +0.74 V
- The following experimental rate data were obtained for a reaction carried out at 25o C:
A(g) + B(g) —-> C(g) + D(g)
- A voltaic cell is made by connecting two half cells represented by half equations below:
Sn2+(aq) + 2e- –>Sn(s)Eo=-0.14 V
Fe3+(aq)+e- –> Fe2+ (aq)Eo=+0.77 V
- Which statement is correct about this voltaic cell?
(a) Fe2+ is oxidised and the voltage of the cell is -0.91 V. (b) Sn is oxidised and the voltage of the cell is 0.91 V. (c) Fe2+ is oxidised and the voltage of the cell is 0.91 V. (d) Sn is oxidised and the voltage of the cell is 0.63 V.
Answer: to be updated
- What are the orders with respect to A(g) and B(g)?
Answer: To be updated
- Which of the following aqueous solution will have highest boiling point?
(a) 1.0 M KCl
(b) 1.0 M K 2 SO4
(c) 2.0 M KCl
(d) 2.0 M K2SO4
- Amides can be converted into amines by the reaction named
(a) Hoffmann degradation
(b) Ammonolysis
(c) Carbylamine
(d) Diazotisation
Answer: (a) Hoffmann degradation
- Which of the following would not be a good choice for reducing nitrobenzene to aniline?
(c) Fe and HCl
(d) Sn and HCl
Answer: (a) LiA l H 4
- Which property of transition metals enables them to behave as catalysts?
(a) High melting point
(b) High ionisation enthalpy
(c) Alloy Formation
(d) Variable oxidation states
Answer: (d) Variable oxidation states
- Which of the following statements is not true about glucose?
(a) It is an aldohexose.
(b) On heating with HI it forms n-hexane.
(c) It is present in pyranose form.
(d) It gives 2,4 DNP test.
Answer: (d) It gives 2,4 DNP test.
- Which of the following alcohols will not undergo oxidation?
(a) Butanol
(b) Butan-2-ol
(c) 2-Methylbutan-2-ol
(d) 3-Methylbutan-2-ol
Answer: ( d) 3-Methylbutan-2-ol
- Four half reactions I to IV are shown below:
- 2Cl- —> Cl2 + 2e-
- 4OH- —-> O2 + 2H2O + 2e-
- Na+ + e- —-> Na
- 2H+ + 2e- —-> H2
Which two of these reactions are most likely to occur when concentrated brine is electrolysed?
(a) I and III
(b) I and IV
(c) II and III
(d) II and IV
For the questions 15 to 18 , two statements are given – one labeled as Assertion (A) and the other labelled as Reason (R ). Select the correct answer to these questions from the codes (a), (b), (c ), and (d) as given below:
(a) Both Assertion (A) and Reason (R ) are true and (R) is the correct explanation of the Assertion (A). (b) Both Assertion (A) and Reason (R) are true, but Reason (R) is not the correct explanation of the Assertion (A). (c) Assertion (A) is true but Reason (R) is false. (d) Assertion (A) is false, but Reason (R) is true.
- Assertion (A) : Vitamin C cannot be stored in our body.
Reason (R) : Vitamin C is fat soluble and is excreted from the body in urine. Answer: (c) Assertion (A) is true but Reason (R) is false
- Assertion (A) : The half life of a reaction is the time in which the concentration of the reactant is reduced to one half of its initial concentration.
Reason (R) : In first order kinetics when concentration of reactant is doubled, its half life is doubled.
- Assertion (A) : Bromination of benzoic acid gives m-bromobenzoic acid.
Reason (R) : Carboxyl group increases the electron density at the meta position. Answer: (a) Both Assertion (A) and Reason (R ) are true and (R) is the correct explanation of the Assertion (A).
- Assertion (A): EDTA is a hexadentate ligand.
Reason (R) : EDTA has 2 nitrogen and 4 oxygen donor atoms. Answer: (a) Both Assertion (A) and Reason (R ) are true and (R) is the correct explanation of the Assertion (A).
CBSE Class 12 Chemistry Exam Paper Analysis – Last year
We surveyed students to find out what they thought of the CBSE Class 12 Chemistry Paper Review 2024. The overall paper was simple to moderate. The questions were straightforward, and nothing was outside of the syllabus. There were some math problems that tested students’ analytical abilities, but everything else was typical in terms of complexity.
Section A, which consisted of MCQs, was the top-scoring component of the exam, according to our subject experts. For your convenience, the answer key for the Class 12 Chemistry test is provided in the sections below.
Class 12 Chemistry Question Paper PDF of Last year
After Completion of the Exam, here we have provided the CBSE Class 12 Chemistry Question Papers Pdf of last year. Candidates may download Class 12 Chemistry Question Papers and analyze the Class 12 Chemistry Answer key to calculate their expected scores.
Class 12 Chemistry Answer Key- Previous Year’s Repeated Questions
Q.1: Arrange the following compounds in the increasing order of their property indicated .
i) Acetaldehyde, Benzaldehyde, Acetophenone, Acetone(Reactivity towards HCN)
Answer. Acetaldehyde<Acetone<Benzaldehyde<Acetophenone
iii) CH3CH2OH, CH3CHO, CH3COOH (Boiling point)
Answer. CH3COOH<CH3CH2OH<CH3CHO
Q.2: In a plot of m against the square root of concentration (C12) for strong and weak electrolytes, the value of limiting molar conductivity of a weak electrolyte cannot be obtained graphically. Suggest a way to obtain this value. Also state the related law, if any.
Yes, we can do it by Kohlrausch’s law.
Kohlrausch’s law: a statement in physical chemistry: the migration of an ion at infinite dilution is dependent on the nature of the solvent and on the potential gradient but not on the other ions present.
Q.3: Write reasons for the following statements :
(i) Benzoic acid does not undergo Friedel-Crafts reaction.
No, benzoic acid does not undergo Friedel Craft reaction because the carboxylic group is deactivating and the Lewis acid catalyst and carboxylic group are bonded.
(ii) Oxidation of aldehydes is easier than that of ketones.
oxidation of aldenyde is easier than ketone due to presence of H-atoms linked to carbonyl group carbon which is absent in ketones.
Q.4: Write reasons for the following:
(i) Ethylamine is soluble in water whereas aniline is insoluble.
Ethylamine when added to water forms intermolecular H−bonds with water. Hence it is soluble in water. But aniline Can form H−bonding with water to a very small extent due to the presence of a large hydrophobic −C6H5 group. Hence aniline is insoluble in water.
(ii) Amino group is o- and p-directing in aromatic electrophilic substitution reactions, but aniline on nitration gives a substantial amount of m-nitroaniline.
Nitration is carried out in an acidic medium. In an acidic medium, aniline is protonated to give anilinium ion (which is meta-directing).
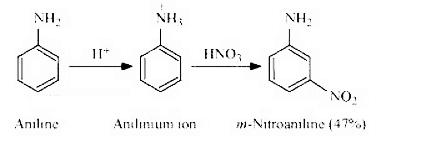
For this reason, aniline on nitration gives a substantial amount of m-nitroaniline.
(iii) Amines behave as nucleophiles.
A nucleophile is a substance that is attracted to, and then attacks, a positive or slightly positive part of another molecule or ion. All amines contain an active lone pair of electrons on the very electronegative nitrogen atom. It is these electrons that are attracted to positive parts of other molecules or ions.
b) How will you carry out the following conversions :
i) Nitrobenzene to Aniline
Nitrobenzene is reduced to aniline by Sn and concentrated HCl. Instead of Sn, Zn or Fe also can be used. Aniline salt is given from this reaction. Then aqueous NaOH is added to the aniline salt to get released aniline. This reaction is called nitrobenzene reduction.
- Benzene is a clear, colourless, highly flammable and volatile, liquid compound.
- Aniline is a yellowish to brownish oily liquid with a musty fishy odour organic compound.
ii) Ethanamide to Methanamine
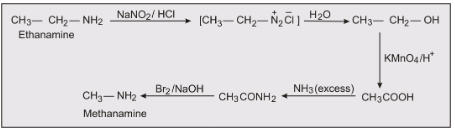
React ethanamine with nitrous acid to form an azo compound, which further reacts with water to form ethanol, which on oxidising gives ethanoic acid. After treating with an excess of ammonia the ethanoic acid becomes ethanamide, which on further reacting with Bomine and a strong base (Hoffmann bromamide degradation reaction) to form methenamine.
iii) Ethanenitrile to Ethanamine
it’s simple reduction of nitriles with lithium aluminium hydride or catalytic hydrogenation produce primary amines. the reaction is
CH3C≡N + H2/Ni OR LiAlH4 → CH3CH2NH2
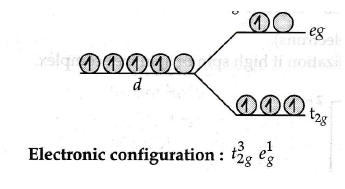
Q.5(a)(i) Write the electronic configuration of d5 on the basis of crystal field splitting theory if Δ0 < P.
When Δo < P, it is weak field and high spin situation’. As a result one electron entered in eg orbital and 3 electrons in t2g.
(ii) [Fe(CN)6]³- is weakly paramagnetic whereas [Fe(CN)6]4- is diamagnetic. Give reason to support this statement [Atomic no. Fe = 26].
Since CN − is a strong field ligand, it causes the pairing of unpaired electrons. Therefore, there is only one unpaired electron left in the d -orbital.
On the other hand, H 2 O is a weak field ligand. Therefore, it cannot cause the pairing of electrons. This means that the number of unpaired electrons is 5.
(iii) Write the number of ions produced from the complex [Co(NH3)6]C12 in solution.
The given complex can be written as [Co(NH3)6]Cl2. Thus, [Co(NH3)6]+ along with two Cl− ions are produced.
Q.(b)(i) Calculate the spin only magnetic moment of the complex [CoF613-. (Atomic no. of Co = 2 7)
Given ion is M 2+ with the atomic no. 27.
(iii) Which out of the two complexes is more stable and why ?
[Fe(H20)6]3+, [Fe(C204)3]3–
[Fe(C204)3]3– is more stable out of two complexes. The central metal ion is Fe3+ and C2O4 2– is negative bi-dentate ligand which forms more stable complex than neutral or monodentate ligand.

Q.6 (i) Which ion amongst the following is colourless and why?
Ti4+, Cr3+, V3+
(Atomic number of Ti = 22, Cr = 24, V = 23)
Ti is colourless because it is having no unpaired e − e- for excitation to higher energy level and it is colourless.
(ii) Why is Mn²+ much more resistant than Fe²+ towards oxidation?
Mn2+ is much more resistant than Fe2+ towards oxidationAs Mn2+ has stable configuration hence it is resistant towards oxidation. while in Fe2+ electronic configuration is 3d6 so it can lose one electron to give stable configuration 3d5.
(iii) Highest oxidation state of a metal is shown in its oxide or fluoride only. Justify the statement.
The highest oxidation state of a metal is exhibited in its oxide or fluoride only. This is because fluorine (F) and oxygen (O)are the most electronegative elements and the highest oxidation state shown by any transition element is +8.
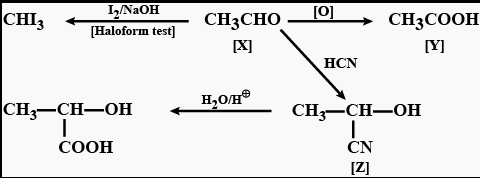
Q.7 A compound ‘A’ (C2H4O) on oxidation gives ‘B’ (C2H4O2). ‘A’ undergoes an Iodoform reaction to give yellow precipitate and reacts with HCN to form the compound ‘C’. ‘C’ on hydrolysis gives 2-hydroxypropanoic acid. Identify the compounds ‘A’, ‘B’ and ‘C’. Write down equations for the reactions involved.
Q.9(a) Write equations involved in the following reactions :
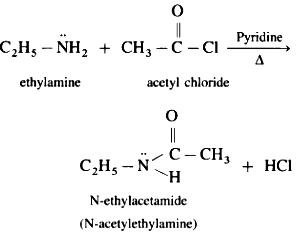
(i) Ethanamine reacts with acetyl chloride.
(ii) Aniline reacts with bromine water at room temperature.
(iii) Aniline reacts with chloroform and ethanolic potassium hydroxide.
When aniline reacts with chloroform and alcoholic K O H it gives an offensive smelling liquid i.e., phenyl isocyanide as product. It is called as isocyanide test. It is given by aliphatic and aromatic primary amines.

Q.9 (b) (i) Write the IUPAC name for the following organic compound: (CH3CH2)2NCH3
( C H 3 C H 2 ) 2 N C H 3 it is a tertiary amine, and the parent chain is ethanamine. IUPAC name is N – Ethyl – N- methyl ethanamine.
(ii) Write the equations for the following:
(I) Gabriel phthalimide synthesis
Gabriel Phthalimide Synthesis Mechanism has 3 steps. The Synthesis is used to get primary amines from primary alkyl halides and is named after the German scientist Siegmund Gabriel.
The reaction has been generalized for applications in the alkylation of sulfonamides and imides & their deprotection in order to obtain amines. Alkylation of ammonia is quite inefficient, therefore it is substituted with phthalimide anion in the Gabriel synthesis.
(II) Hoffmann bromamide degradation
When an amide is treated with bromine in an aqueous or ethanolic solution of sodium hydroxide, degradation of amide takes place leading to the formation of primary amine. This reaction involving degradation of amide and is popularly known as Hoffmann bromamide degradation reaction. The primary amine thus formed contains one carbon less than the number of carbon atoms in that amide.
RCONH 2 +Br 2 + 4NaOH
R-NH 2 + Na 2 CO 3 + 2NaBr + 2H 2 O
Q.11(a) i) Silver atom has completely filled d-orbitals in its ground state, it is still considered to be a transition element. Justify the statement.
Silver (Ag) belongs to group 11 of d-block and its ground state electronic configuration is 4d10 5s1. It shows an oxidation state of +2 in its compounds like AgO and AgF2 in which its electronic configuration is d9 so it is a transition element.
ii) Why are E° values of Mn and Zn more negative than expected?
Negative E° values of Mn2+ and Zn2+ are because of the stabilities of half-filled (3d5 : Mn2+)and fullyfilled (3d10 : Zn2+) configuration respectively. Ni2+ ion has higher E° value due to highest negative enthalpy of hydration.
iii) Why do transition metals form alloys?
Transition metals have very similar atomic sizes. One metal can easily replace the other metal from its lattice to form solid solution (alloy). Transition metals are miscible with one another in the molten state. The molten state solution of two or more transition metals on cooling forms alloy.
Sharing is caring!
When will CBSE class 12 Chemistry Exam takes place?
The CBSE Chemistry Examination is going to take place on 27 February 2024.
How many Sections are there in the class 12 Chemistry Question paper?
Class 12 Chemistry Question paper has 5 sections with 35 questions for 70 marks.
What is the totals marks of class 12 Chemistry Question paper?
The totals marks of the class 12 Chemistry Question paper is 70 marks
Hi, I am Brajesh (M.Tech, MCA), I Professional Educator having 3 years of experience in school education sector. Aim to provide JEE, NEET, CUET, and Other Entrance exams information in a simple way to help students find clarity and confidence. I provide here easily accessible content on Exam Notifications, Syllabus, Admit Cards, and Results.

Join the Conversation
Keep it up the good work
Great article, the information provided to us. Thank you
Sir we need for physics
nice solutions for class 12 chemistry
Your email address will not be published. Required fields are marked *
Save my name, email, and website in this browser for the next time I comment.
Leave a comment
Trending Articles
- CBSE Class 10 Result 2024
- CBSE Class 12th Result 2024
- CGBSE 10th Result 2024
- NEET Question Paper 2024
- NEET Answer Key 2024 All Sets
- NEET Exam Analysis 2024
- NEET Expected Cut Off 2024

CBSE Board Exam 2024
- CBSE Class 10 Syllabus 2024
- CBSE Class 12 Syllabus 2024
- CBSE Previous Year Papers
- CUET Syllabus
- CUET Previous Year paper
- CUET Participating College & Universities
- JEE Main 2024
- JEE Main Syllabus 2024
- JEE Main Exam Analysis 2023
- NEET 2024
- NEET Syllabus 2024
- NEET State wise Cut off
- NEET Rank Predictor
- NEET OMR Sheet
- NEET College Predictor
Recent Posts
Important exams, ncert solutions.
- NCERT Class 12
- NCERT Class 11
- NCERT Class 10
- NCERT Class 9
NCERT Books
School syllabus.
- CBSE Class 12
- CBSE Class 11
- CBSE Class 10
- CBSE Class 9
- JEE Mains 2024
Our Other Websites
- Teachers Adda
- Bankers Adda
- Current Affairs
- Adda Bengali
- Engineers Adda
- Adda Marathi
- Adda School

Get all your queries solved in one single place. We at Adda247 school strive each day to provide you the best material across the online education industry. We consider your struggle as our motivation to work each day.
Download Adda247 App
Follow us on
- Responsible Disclosure Program
- Cancellation & Refunds
- Terms & Conditions
- Privacy Policy
- Share full article
Advertisement
Supported by
Solar Storm Intensifies, Filling Skies With Northern Lights
Officials warned of potential blackouts or interference with navigation and communication systems this weekend, as well as auroras as far south as Southern California or Texas.

By Katrina Miller and Judson Jones
Katrina Miller reports on space and astronomy and Judson Jones is a meteorologist.
A dramatic blast from the sun set off the highest-level geomagnetic storm in Earth’s atmosphere on Friday that is expected to make the northern lights visible as far south as Florida and Southern California and could interfere with power grids, communications and navigations system.
It is the strongest such storm to reach Earth since Halloween of 2003. That one was strong enough to create power outages in Sweden and damage transformers in South Africa.
The effects could continue through the weekend as a steady stream of emissions from the sun continues to bombard the planet’s magnetic field.
The solar activity is so powerful that the National Oceanic and Atmospheric Administration, which monitors space weather, issued an unusual storm watch for the first time in 19 years, which was then upgraded to a warning. The agency began observing outbursts on the sun’s surface on Wednesday, with at least five heading in the direction of Earth.
“What we’re expecting over the next couple of days should be more significant than what we’ve seen certainly so far,” Mike Bettwy, the operations chief at NOAA’s Space Weather Prediction Center, said at a news conference on Friday morning.
For people in many places, the most visible part of the storm will be the northern lights, known also as auroras. But authorities and companies will also be on the lookout for the event’s effects on infrastructure, like global positioning systems, radio communications and even electrical power.
While the northern lights are most often seen in higher latitudes closer to the North Pole, people in many more parts of the world are already getting a show this weekend that could last through the early part of next week.

As Friday turned to Saturday in Europe, people across the continent described skies hued in a mottling of colors.
Alfredo Carpineti , an astrophysicist, journalist and author in North London, saw them with his husband from the rooftop of their apartment building.
“It is incredible to be able to see the aurora directly from one’s own backyard,” he said. “I was hoping to maybe catch a glimpse of green on the horizon, but it was all across the sky in both green and purple.”
Here’s what you need to know about this weekend’s solar event.
How will the storm affect people on Earth?
A geomagnetic storm watch or warning indicates that space weather may affect critical infrastructure on or orbiting near Earth. It may introduce additional current into systems, which could damage pipelines, railroad tracks and power lines.
According to Joe Llama, an astronomer at Lowell Observatory, communications that rely on high frequency radio waves, such as ham radio and commercial aviation , are most likely to suffer. That means it is unlikely that your cellphone or car radio, which depend on much higher frequency radio waves, will conk out.
Still, it is possible for blackouts to occur. As with any power outage, you can prepare by keeping your devices charged and having access to backup batteries, generators and radio.
The most notable solar storm recorded in history occurred in 1859. Known as the Carrington Event, it lasted for nearly a week, creating aurora that stretched down to Hawaii and Central America and impacting hundreds of thousands of miles of telegraph lines.
But that was technology of the 19th century, used before scientists fully understood how solar activity disrupted Earth’s atmosphere and communication systems.
“That was an extreme level event,” said Shawn Dahl, a forecaster at NOAA’s Space Weather Prediction Center. “We are not anticipating that.”
Unlike tornado watches and warnings, the target audience for NOAA’s announcements is not the public.
“For most people here on planet Earth, they won’t have to do anything,” said Rob Steenburgh, a space scientist at NOAA’s Space Weather Prediction Center.
The goal of the announcements is to give agencies and companies that operate this infrastructure time to put protection measures in place to mitigate any effects.
“If everything is working like it should, the grid will be stable and they’ll be able to go about their daily lives,” Mr. Steenburgh said.

Will I be able to see the northern lights?
It is possible that the northern lights may grace the skies this week over places that don’t usually see them. The best visibility is outside the bright lights of cities.
Clouds or stormy weather could pose a problem in some places. But if the skies are clear, even well south of where the aurora is forecast to take place, snap a picture or record a video with your cellphone. The sensor on the camera is more sensitive to the wavelengths produced by the aurora and may produce an image you can’t see with the naked eye.
Another opportunity could be viewing sunspots during the daytime, if your skies are clear. As always, do not look directly at the sun without protection. But if you still have your eclipse glasses lying around from the April 8 event, you may try to use them to try to spot the cluster of sunspots causing the activity.
How strong is the current geomagnetic storm?
Giant explosions on the surface of the sun, known as coronal mass ejections, send streams of energetic particles into space. But the sun is large, and such outbursts may not cross our planet as it travels around the star. But when these particles create a disturbance in Earth’s magnetic field, it is known as a geomagnetic storm.
NOAA classifies these storms on a “G” scale of 1 to 5, with G1 being minor and G5 being extreme. The most extreme storms can cause widespread blackouts and damage to infrastructure on Earth. Satellites may also have trouble orienting themselves or sending or receiving information during these events.
The current storm is classified as G5, or “extreme.” It is caused by a cluster of sunspots — dark, cool regions on the solar surface — that is about 16 times the diameter of Earth. The cluster is flaring and ejecting material every six to 12 hours.
“We anticipate that we’re going to get one shock after another through the weekend,” said Brent Gordon, chief of the space weather services branch at NOAA’s Space Weather Prediction Center.
Why is this happening now?
The sun’s activity ebbs and flows on an 11-year cycle, and right now, it is approaching a solar maximum. Three other severe geomagnetic storms have been observed so far in the current activity cycle, which began in December 2019, but none were predicted to cause effects strong enough on Earth to warrant a watch or warning announcement.
The cluster of sunspots generating the current storm is the largest seen in this solar cycle, NOAA officials said. They added that the activity in this cycle has outperformed initial predictions .
More flares and expulsions from this cluster are expected, but because of the sun’s rotation the cluster will be oriented in a position less likely to affect Earth. In the coming weeks, the sunspots may appear again on the left side of the sun, but it is difficult for scientists to predict whether this will cause another bout of activity.
“Usually, these don’t come around packing as much of a punch as they did originally,” Mr. Dahl said. “But time will tell on that.”
Jonathan O’Callaghan contributed reporting from London.
An earlier version of this article misstated the radio frequencies used by cellphones and car radios. They are higher frequencies, not low.
How we handle corrections
Katrina Miller is a science reporting fellow for The Times. She recently earned her Ph.D. in particle physics from the University of Chicago. More about Katrina Miller
Judson Jones is a meteorologist and reporter for The Times who forecasts and covers extreme weather. More about Judson Jones
What’s Up in Space and Astronomy
Keep track of things going on in our solar system and all around the universe..
Never miss an eclipse, a meteor shower, a rocket launch or any other 2024 event that’s out of this world with our space and astronomy calendar .
A dramatic blast from the sun set off the highest-level geomagnetic storm in Earth’s atmosphere, making the northern lights visible around the world .
With the help of Google Cloud, scientists who hunt killer asteroids churned through hundreds of thousands of images of the night sky to reveal 27,500 overlooked space rocks in the solar system .
A celestial image, an Impressionistic swirl of color in the center of the Milky Way, represents a first step toward understanding the role of magnetic fields in the cycle of stellar death and rebirth.
Scientists may have discovered a major flaw in their understanding of dark energy, a mysterious cosmic force . That could be good news for the fate of the universe.
Is Pluto a planet? And what is a planet, anyway? Test your knowledge here .

MBSE HSSLC Result 2024 Date, Time: Mizoram Board Class 12th results releasing on May 21 at 12 noon
M izoram Board of School Education has announced the MBSE HSSLC Result 2024 Date, Time. The Mizoram Board Class 12th results for Science, Commerce and Arts stream will be declared on May 21 at 12 noon. Candidates who have appeared for the Class 12 board examination can check the scores on the official website of MBSE at mbse.edu.in. The result link will also be available on mbseonline.com.
Apart from the official website, the results can also be obtained by appeared candidates on whatsapp numbers- 9863883041 and 9863722521 during office hours.
All the candidates who have appeared for the examination can check the scores by following the steps given below.
- Visit the official website of MBSE at mbse.edu.in.
- Click on MBSE HSSLC Result 2024 link available on the home page.
- Enter the login details and click on submit.
- Once done, fill the application form.
- Make the payment of application fee.
- Click on submit and download the page.
- Keep a hard copy of the same for further need.
This year Mizoram Class 12 board examination was conducted from February 28 to March 28, 2024. The examination was conducted in single shift- from 10 am to 1 pm. For more related details candidates can check the official website of MBSE.
Read more news like this on HindustanTimes.com


IMAGES
VIDEO
COMMENTS
The theory paper for class 7 CBSE English is divided into 3 sections the first one is for 20 marks that test the comprehension reading ability of the students and the questions are to be answered based on the given passage or a poem. The second section is for 30 marks which are to test the grammar knowledge and writing skills of the students.
Students can get the latest CBSE Sample Paper for Class 7 for the exam on this page. To help students prepare effectively for the exam, we have compiled sample papers for Maths, Science, English and Social Science subjects. Upon clicking on the respective link provided in the table above, students can easily download the sample papers.
List of 7th Standard Essay Topics & Ideas for Kids in English. Class 7 Essay Topics relating to different categories exist here. Students of 7th grade are advised to Read and Practice all covered English Essay Topics for 7th Std for enhancing your analytical & writing skills. For aiding in your practice sessions of essay writings, we have ...
The 7th class final examinations are held according to the school-issued schedule by the administration. Students looking for 7th class past papers are welcome to use this site. For your convenience, we have made the source of past papers available on our website, ilmkidunya. You can visit the website any time and get the past papers there.
Here are some exercises that Class 7 students can use to practice their essay-writing skills: 1. Choose a topic and write a five-paragraph essay using the basic essay format. 2. Read a news article and write an essay summarizing the main points and providing your opinion on the topic. 3.
2022 Grade 7 ELA Test Text Complexity Metrics for Released Questions Available. Selecting high-quality, grade-appropriate passages requires both objective text complexity metrics and expert judgment. For the Grades 3-8 assessments based on the New York State P-12 Learning Standards for English Language Arts, both quantitative and qualitative ...
If the student writes only a personal response and makes no reference to the text(s), the response can be scored no higher than a 1. Responses totally unrelated to the topic, illegible, or incoherent should be given a 0. A response totally copied from the text(s) with no original student writing should be scored a 0.
2019 Grade 7 ELA Test Text Complexity Metrics for Released Questions Available on EngageNY. Selecting high-quality, grade-appropriate passages requires both objective text complexity metrics and expert judgment. For the Grades 3-8 assessments based on the New York State P-12 Learning Standards for English Language Arts, both quantitative and ...
Our completely free Common Core: 7th Grade English Language Arts practice tests are the perfect way to brush up your skills. Take one of our many Common Core: 7th Grade English Language Arts practice tests for a run-through of commonly asked questions. You will receive incredibly detailed scoring results at the end of your Common Core: 7th ...
FSA ELA Writing Practice Test. The purpose of these practice test materials is to orient teachers and students to the types of passages and prompts on FSA ELA Writing tests. Each spring, students in grades 4-10 are administered one text-based writing prompt for the FSA English Language Arts test. Students will respond to either an informative ...
15 Another factor in choosing a capital is the city's size and economic health. While D.C. has a sizable population, there are cities in the United States with far more people, businesses, and growth opportunities. New York City, Los Angeles, Chicago, Houston, and Phoenix are the five most populous cities in the country.
All practice test items represent the appropriate grade level/content standards—however, the practice test may contain item types that no longer appear on the operational assessment. Published under contract with the Tennessee Department of Education by Questar Assessment Inc., 5550 Upper 147th Street West, Minneapolis, MN 55124.
A. As basketball star Charles Barkley stated in a famous advertising campaign for Nike, he was paid to dominate on the basketball court, not to raise your kids. Many celebrities do consider themselves responsible for setting a good example and create non-profit organizations through which they can benefit youths. B.
1) Unity: The essay should deal with the main subject and all parts of it should be clearly linked with that subject. 2) Coherence: There should be a logical sequence of thought. This requires a logical relationship between ideas, sentences and paragraphs. 3) Relevance: Unimportant information should not be included.
Testpapers is your companion in paving a successful academic path in Grade 7. Largest library of grade 7 CAPS exam papers for South African students, available in both English & Afrikaans. Over 300+ exam papers & memos for 11 subjects. All our exam papers are 100% free to access.
7th grade. 9 units · 119 skills. Unit 1. Proportional relationships. Unit 2. Rates and percentages. Unit 3. Integers: addition and subtraction. Unit 4. ... Course challenge Test your knowledge of the skills in this course. Start Course challenge. Community questions. Our mission is to provide a free, world-class education to anyone, anywhere.
PHYSICAL EDUCATION. Join Kenya's Largest Teachers Telegram Group with Over 80K Teachers FORM 1-4 CLASS 7-8 GRADE 1-6 PP1-PP2 KASNEB PTE. Download all the 2023 Grade 7 Exams for Term 1, Term 2, and Term 3.
Released Questions. 2021. New York State administered the English Language Arts Tests in April 2021 and is now making the questions from Session 1 of these tests available for review and use. Only Session 1 was required in 2021. THE STATE EDUCATION DEPARTMENT / THE UNIVERSITY OF THE STATE OF NEW YORK / ALBANY, NY 12234.
grade 7 agric term 2 opener .pdf grade 7 term 2 performing arts schemes.docx (3).pdf grade 7 bus term 2 opener exam .pdf grade 7 comp term 2 opener exam .pdf grade 7 int sci term 2 opener exam .pdf grade 7 eng term 2 opener .pdf grade 7 fre term 2 opener .pdf grade 7 term 2 physical education schemes (3).pdf grade 7 cre term 2 opener exam .pdf ...
CBSE Class 10 Maths Exam 2024: The CBSE Class 10 results are out and students who got the passing marks are happy.Around 93% students got the passing score and less 7% are still in the race to ...
CBSE Class 7 Science Sample Paper Set 8. Students can look at the Summative assessment question paper of CBSE Class 7 Science below in the table. We have provided the SA1 and SA2 Science sample papers for students' practice. These CBSE Sample Papers will also boost their exam preparation and help them score more marks in the exam.
Worry not, the recent CBSE Class 10 board papers will help you in that. On March 11, 2024, the CBSE 10th Mathematics exam was conducted for 041 and 241 subject codes. The examination was scheduled ...
The question paper for the shift 1 and shift 2 exam paper of May 15 is given below. CUET UG Question Paper 2024. The CUET UG 2024 exam is being held in hybrid mode for the first time. The examination for the online mode will be held on computers while the offline mode examination will be organized through conventional method on pen and paper.
They also pick up skills like recitation, pronunciation, confidence, etc. Hence, in this blog, we bring you 7 Interesting and Fun English Stories For Class 3. Here, you will find some great stories for kids. Let's explore them now! Table of Contents [ hide] 1 1. The Bear and the Two Friends. 2 2. The Miser and His Gold.
2023 Grade 7 ELA Test Text Complexity Metrics for. Released Questions Available. Selecting high-quality, grade-appropriate passages requires both objective text complexity metrics and expert judgment. the For Grades 3-8 assessments based on the New York State P-12 Next Generation Learning Standards for English Language Arts, both quantitative ...
To apply for NABARD Grade A Recruitments, candidates must meet the criteria listed in the Grade A 2023 exam notification: 1) Age Requirement (as of September 1, 2023) You have to be between the ages of 21 and 30 on September 1, 2023. In other words, neither 02/09/1993 nor 01/09/2002 can be your birthdate.
CBSE Class 12 Chemistry Answer Key Set 3 Last year. 1, A compound CaCl2.6h2O undergoes complete dissociation in water, The Van't Hoff Factor 'i' is: a) 9, b) 6, c)3, d) . 4. Answer: c) 3. 2. For a Zero order reaction of type A →products, the rate equation may be expressed as: Answer: Option (a) 3.
For people in many places, the most visible part of the storm will be the northern lights, known also as auroras. But authorities and companies will also be on the lookout for the event's ...
The Mizoram Board Class 12th results for Science, Commerce and Arts stream will be declared on May 21 at 12 noon. Candidates who have appeared for the Class 12 board examination can check the ...